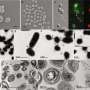
Special Nanotubes May Be Used as a Vehicle for Treating Neurodegenerative Disorders

(PhysOrg.com) -- Electrical engineering researchers at the University of Arkansas have demonstrated that magnetic nanotubes combined with nerve growth factor can enable specific cells to differentiate into neurons. The results from in vitro studies show that magnetic nanotubes may be exploited to treat neurodegenerative disorders such as Parkinson’s disease and Alzheimer’s disease because they can be used as a delivery vehicle for nerve growth factor.
“Due to their structure and properties, magnetic nanotubes are among the most promising candidates of multifunctional nanomaterials for clinical diagnostic and therapeutic applications,” said Jining Xie, research assistant professor and lead author of the study. “We’re excited about these results specifically and the overall promise of functionalized nanotubes to treat patients with these debilitating conditions.”
Xie, Linfeng Chen, senior research associate in the Center for Wireless Nano-, Bio- and Info-Tech Sensors and Systems, and researchers from Arkansas State University worked with rat pheochromocytoma, otherwise known as PC12 cells. PC12 cells were chosen because they require nerve growth factor - a small, secreted protein that helps nerve cells survive - to differentiate into neurons.
The researchers knew that any sign of nerve growth would indicate interactions between the PC12 cells and the nanotubes. They observed neurite growth, specifically filopodia - slender projections that extend from the leading edge of migrating cells - extruding from neurite growth cones toward the nanotubes incorporated with nerve growth factor.
“Microscopic observations showed filopodia extending from the growth cones were in close proximity to the nanotubes, at time appearing to reach out toward or into them,” Xie said.
The results did not show any abnormal toxicity from the nanotubes.
The human nervous system depends upon a complex network of neurons, or nerve cells, tied to each other by synapses. The synaptic connections occur through neurites, which are immature or developing neurons. When these connections fail, the nervous system does not function properly, eventually leading to injury or disease. As Xie mentioned, the researchers hope that functionalized nanotubes can help restore or repair damaged nerve cells.
Xie collaborates with Vijay Varadan, distinguished professor of electrical engineering and director of the Center for Wireless Nano-, Bio- and Info-Tech Sensors and Systems, which is supported by the National Science Foundation’s Experimental Program to Stimulate Competitive Research. Another collaborator was Malathi Srivatsan, associate professor of biology at Arkansas State University in Jonesboro, Ark.
Varadan holds the College of Engineering’s Twenty-First Century Endowed Chair in Nano- and Bio-Technologies and Medicine and the college’s Chair in Microelectronics and High Density Electronics. In addition to his position as director of the above center, he directs the university’s High Density Electronics Center. Varadan is also a professor of neurosurgery in the College of Medicine at the University of Arkansas for Medical Sciences in Little Rock, Ark.
The researchers’ findings were published in Nanotechnology, an Institute of Physics Publishing journal. An online version of the article is available at www.iop.org/EJ/abstract/-searc … 57-4484/19/10/105101 .
Provided by University of Arkansas