Scientists peel away the mystery behind gold's catalytic prowess
Few materials have exercised as much of a hold on the human imagination, or on human history, as has gold. But for all of its popular uses – money, medals, jewelry and more – gold's potential as a catalyst lay hidden until the 1980s, when Masatake Haruta and Graham Hutchings independently discovered that gold, which had long been considered inactive, could be an extraordinarily good catalyst. Haruta demonstrated the low-temperature oxidation of CO and Hutchings the hydrochlorination of acetylene to vinyl chloride.
Gold particles measuring less than 5 nanometers in diameter possess a high level of catalytic activity when they are deposited on metal-oxide supports, Haruta learned. One nanometer (nm) is equal to one one-billionth of a meter, or about the width of five atoms.
In particular, Haruta found that gold nanoparticles are effective at catalyzing the critical conversion of toxic carbon monoxide (CO) into more benign carbon dioxide (CO2) at room temperature and even at temperatures as low as -76 degrees C. CO oxidation is vital to firefighters and others who must enter burning buildings, and it is also critical to the protection of hydrogen fuel cells from CO contamination.
In the two decades since Haruta's discovery, scientists have sought to determine exactly how gold nanoparticles function as catalysts.
Now, researchers from Lehigh University in Bethlehem, Pa., and Cardiff University in the UK believe they have pinpointed the active species at which the critical oxidation reaction occurs when gold is supported on iron oxide.
In an article to be published in Science, the premier scientific journal in the U.S., researchers from Lehigh University in Bethlehem, Pa.; Cardiff University in Wales, and the National Institute of Standards and Technology (NIST) report that bilayer clusters measuring about one-half nanometer in diameter and containing only about 10 gold atoms are responsible for triggering the CO oxidation reaction.
The researchers, using aberration-corrected transmission electron microscopy capable of resolving single gold atoms, also report that a simple change in preparation – the drying of the catalyst in flowing rather than static air – helps impart to the gold its catalytic capability.
The article, titled "Identification of Active Gold Nanoclusters on Iron Oxide Supports for CO Oxidation," is scheduled to be published in the Sept. 5 issue of Science.
Its authors are Christopher Kiely, director of the Nanocharacterization Laboratory in Lehigh's Center for Advanced Materials and Nanotechnology; Graham Hutchings, Albert Carley and Philip Landon of Cardiff's School of Chemistry; and Andrew Herzing of NIST's Surface and Microanalysis Science Division. Herzing earned a Ph.D. from Lehigh in 2006.
Hutchings and Kiely have collaborated since 1989 and have worked together on gold catalysts since 2000. In this project, Hutchings' group carried out the fabrication and catalytic testing of the gold nanoparticles, and the characterization of the catalyst using x-ray photoelectron spectroscopy (XPS). Kiely's group then used Lehigh's aberration-corrected 2200 JEOL scanning transmission electron microscope (STEM) to examine the gold's nanostructure. Lehigh is currently the only university in the world with two aberration-corrected electron microscopes, which are the world's most powerful instruments for chemical analysis.
The researchers compared two groups of gold nanoparticles. One, dried in static air, was what scientists call a "dead" catalyst with little or no catalytic activity. The other group, dried with flowing air, was a 100-percent-active catalyst for CO oxidation.
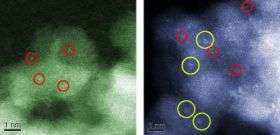
On the inactive catalyst, Herzing saw two types of gold species – particles larger than 1 nm in size and individual atoms scattered about on the iron-oxide support. On the 100-percent-active catalyst, he found a third species – clusters of 8 to 12 gold atoms arranged in two layers measuring about 0.5 nm in dimension.
"This was the clue that enabled us to identify the tiny bilayer clusters as the important species in the catalytic reaction," said Kiely. "It turns out that only about 2 percent of the gold deposited on the support ended up in this particular type of cluster.
"We then deactivated the catalyst by various heat treatments and found that we could correlate the loss of the clusters with the loss of activity. This gives us strong evidence that the active species in the catalyst are the tiny bilayer clusters.
"We believe we have obtained the first conclusive evidence that bilayer clusters are occurring in a real gold catalyst, that they are the key species on that catalyst, and that their presence or absence correlates with the ability or failure of the catalyst to perform CO oxidation."
Before Lehigh's acquisition of the aberration-corrected electron microscopes in 2004, Kiely and Hutchings were able to see the larger gold particles, but not the individual atoms or bilayer clusters of atoms.
"At that time, when we compared the dead catalyst and the active catalyst," said Kiely, "both looked the same. The new microscopes have opened up a new window allowing us to see what is really going on."
The aberration-corrected STEM enabled Herzing and Kiely to use a microscopy technique called high-angle annular dark-field imaging, which requires an extremely fine, 1-angstrom-wide beam of electrons to obtain a scanned image of a specimen. An angstrom is equal to one-tenth of a nanometer.
Kiely said the gold catalysts could find a potential application in the protective masks capable of converting CO to CO2 that are worn by firefighters and others exposed to high levels of CO. Another application is to fuel cells that are vulnerable to poisoning by the CO that is present in the hydrogen fuel stream.
Gold catalysts are also being explored for their effectiveness in catalyzing the reaction that is used to steam-reform methane into hydrogen.
The article by Kiely, Hutchings and their collaborators is the second by the group to be published by Science in the past two years. The first, published in 2006, reported on the use of gold-palladium nanoparticles to catalyze the conversion of alcohols into aldehydes, the chemical process that is important in the synthesis of some spices and perfumes.
In 2005, the group reported in Nature that the selective oxidation processes used to make compounds contained in agrochemicals, pharmaceuticals and other chemical products could be accomplished more cleanly and more efficiently with gold nanoparticle catalysts.
Source: Lehigh University