Beyond batteries: Storing power in a sheet of nanocomposite paper
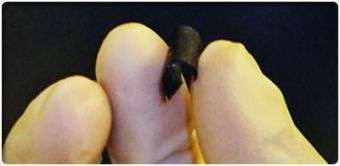
Researchers at Rensselaer Polytechnic Institute have developed a new energy storage device that easily could be mistaken for a simple sheet of black paper.
The nanoengineered battery is lightweight, ultra thin, completely flexible, and geared toward meeting the trickiest design and energy requirements of tomorrow’s gadgets, implantable medical equipment, and transportation vehicles.
Along with its ability to function in temperatures up to 300 degrees Fahrenheit and down to 100 below zero, the device is completely integrated and can be printed like paper. The device is also unique in that it can function as both a high-energy battery and a high-power supercapacitor, which are generally separate components in most electrical systems. Another key feature is the capability to use human blood or sweat to help power the battery.
Details of the project are outlined in the paper “Flexible Energy Storage Devices Based on Nanocomposite Paper” published Aug. 13 in the Proceedings of the National Academy of Sciences.
The semblance to paper is no accident: more than 90 percent of the device is made up of cellulose, the same plant cells used in newsprint, loose leaf, lunch bags, and nearly every other type of paper.
Rensselaer researchers infused this paper with aligned carbon nanotubes, which give the device its black color. The nanotubes act as electrodes and allow the storage devices to conduct electricity. The device, engineered to function as both a lithium-ion battery and a supercapacitor, can provide the long, steady power output comparable to a conventional battery, as well as a supercapacitor’s quick burst of high energy.
The device can be rolled, twisted, folded, or cut into any number of shapes with no loss of mechanical integrity or efficiency. The paper batteries can also be stacked, like a ream of printer paper, to boost the total power output.
“It’s essentially a regular piece of paper, but it’s made in a very intelligent way,” said paper co-author Robert Linhardt, the Ann and John H. Broadbent Senior Constellation Professor of Biocatalysis and Metabolic Engineering at Rensselaer.
“We’re not putting pieces together – it’s a single, integrated device,” he said. “The components are molecularly attached to each other: the carbon nanotube print is embedded in the paper, and the electrolyte is soaked into the paper. The end result is a device that looks, feels, and weighs the same as paper.”
The creation of this unique nanocomposite paper drew from a diverse pool of disciplines, requiring expertise in materials science, energy storage, and chemistry. Along with Linhardt, authors of the paper include Pulickel M. Ajayan, professor of materials science and engineering, and Omkaram Nalamasu, professor of chemistry with a joint appointment in materials science and engineering. Senior research specialist Victor Pushparaj, along with postdoctoral research associates Shaijumon M. Manikoth, Ashavani Kumar, and Saravanababu Murugesan, were co-authors and lead researchers of the project. Other co-authors include research associate Lijie Ci and Rensselaer Nanotechnology Center Laboratory Manager Robert Vajtai.
The researchers used ionic liquid, essentially a liquid salt, as the battery’s electrolyte. It’s important to note that ionic liquid contains no water, which means there’s nothing in the batteries to freeze or evaporate. “This lack of water allows the paper energy storage devices to withstand extreme temperatures,” Kumar said.
Along with use in small handheld electronics, the paper batteries’ light weight could make them ideal for use in automobiles, aircraft, and even boats. The paper also could be molded into different shapes, such as a car door, which would enable important new engineering innovations.
“Plus, because of the high paper content and lack of toxic chemicals, it’s environmentally safe,” Shaijumon said.
Paper is also extremely biocompatible and these new hybrid battery/supercapcitors have potential as power supplies for devices implanted in the body. The team printed paper batteries without adding any electrolytes, and demonstrated that naturally occurring electrolytes in human sweat, blood, and urine can be used to activate the battery device.
“It’s a way to power a small device such as a pacemaker without introducing any harsh chemicals – such as the kind that are typically found in batteries – into the body,” Pushparaj said.
The materials required to create the paper batteries are inexpensive, Murugesan said, but the team has not yet developed a way to inexpensively mass produce the devices. The end goal is to print the paper using a roll-to-roll system similar to how newspapers are printed.
“When we get this technology down, we’ll basically have the ability to print batteries and print supercapacitors,” Ajayan said. “We see this as a technology that’s just right for the current energy market, as well as the electronics industry, which is always looking for smaller, lighter power sources. Our device could make its way into any number of different applications.”
The team of researchers has already filed a patent protecting the invention. They are now working on ways to boost the efficiency of the batteries and supercapacitors, and investigating different manufacturing techniques.
Source: Rensselaer Polytechnic Institute





















