Electric fields remove nanoparticles from blood with ease
Engineers at the University of California, San Diego developed a new technology that uses an oscillating electric field to easily and quickly isolate drug-delivery nanoparticles from blood. The technology could serve as a general tool to separate and recover nanoparticles from other complex fluids for medical, environmental, and industrial applications.
Nanoparticles, which are generally one thousand times smaller than the width of a human hair, are difficult to separate from plasma, the liquid component of blood, due to their small size and low density. Traditional methods to remove nanoparticles from plasma samples typically involve diluting the plasma, adding a high concentration sugar solution to the plasma and spinning it in a centrifuge, or attaching a targeting agent to the surface of the nanoparticles. These methods either alter the normal behavior of the nanoparticles or cannot be applied to some of the most common nanoparticle types.
"This is the first example of isolating a wide range of nanoparticles out of plasma with a minimum amount of manipulation," said Stuart Ibsen, a postdoctoral fellow in the Department of NanoEngineering at UC San Diego and first author of the study published October in the journal Small. "We've designed a very versatile technique that can be used to recover nanoparticles in a lot of different processes."
This new nanoparticle separation technology will enable researchers—particularly those who design and study drug-delivery nanoparticles for disease therapies—to better monitor what happens to nanoparticles circulating in a patient's bloodstream. One of the questions that researchers face is how blood proteins bind to the surfaces of drug-delivery nanoparticles and make them less effective. Researchers could also use this technology in the clinic to determine if the blood chemistry of a particular patient is compatible with the surfaces of certain drug-delivery nanoparticles.
"We were interested in a fast and easy way to take these nanoparticles out of plasma so we could find out what's going on at their surfaces and redesign them to work more effectively in blood," said Michael Heller, a nanoengineering professor at the UC San Diego Jacobs School of Engineering and senior author of the study.

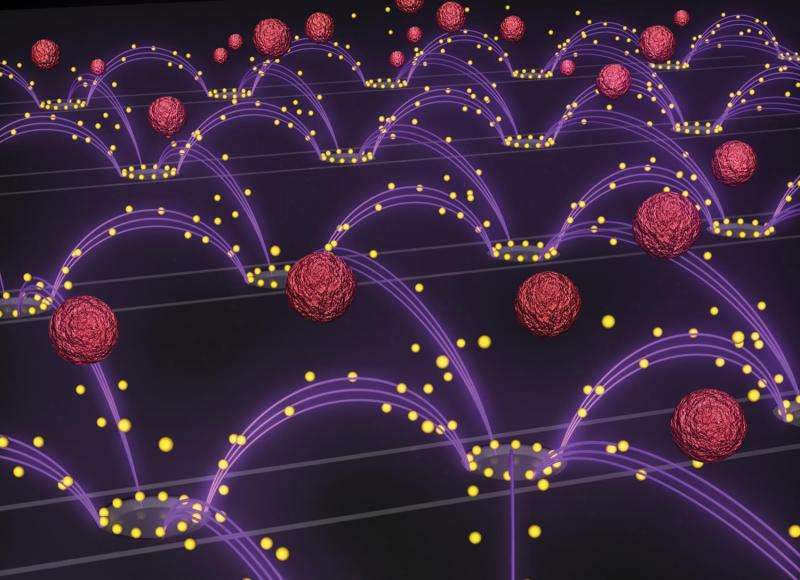
The device used to isolate the drug-delivery nanoparticles was a dime-sized electric chip manufactured by La Jolla-based Biological Dynamics, which licensed the original technology from UC San Diego. The chip contains hundreds of tiny electrodes that generate a rapidly oscillating electric field that selectively pulls the nanoparticles out of a plasma sample. Researchers inserted a drop of plasma spiked with nanoparticles into the electric chip and demonstrated nanoparticle recovery within 7 minutes. The technology worked on different types of drug-delivery nanoparticles that are typically studied in various labs.
The breakthrough in the technology relies on designing a chip that can work in the high salt concentration of blood plasma. The chip's ability to pull the nanoparticles out of plasma is based on differences in the material properties between the nanoparticles and plasma components. When the chip's electrodes apply an oscillating electric field, the positive and negative charges inside the nanoparticles reorient themselves at a different speed than the charges in the surrounding plasma. This momentary imbalance in the charges creates an attractive force between the nanoparticles and the electrodes. As the electric field oscillates, the nanoparticles are continually pulled towards the electrodes, leaving the rest of the plasma behind. Also, the electric field is designed to oscillate at just the right frequency: 15,000 times per second.
"It's amazing that this method works without any modifications to the plasma samples or to the nanoparticles," said Ibsen.

More information: "Recovery of Drug Delivery Nanoparticles from Human Plasma Using an Electrokinetic Platform Technology," by Stuart Ibsen, Avery Sonnenberg, Carolyn Schutt, Rajesh Mukthavaram, Yasan Yeh, Inanc Ortac, Sareh Manouchehri, Santosh Kesari, Sadik Esener, and Michael J. Heller. The paper was published in the Oct. 14, 2015 issue of the journal Small. onlinelibrary.wiley.com/doi/10 … /smll.201570233/full
Journal information: Small
Provided by University of California - San Diego