Key adjustment enables parasite shape-shifting
Crafty parasites frequently undergo dramatic shape changes during their life cycles that enable them to adapt to different living conditions and thrive. But these transformations might not be as difficult as they appear, according to a study in The Journal of Cell Biology.
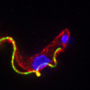
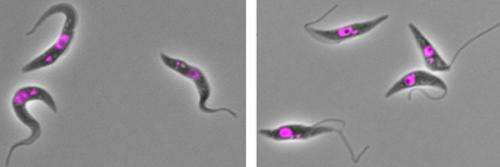
African "sleeping sickness" is a disease caused by a species of parasite known as Trypanosoma brucei that is transmitted by the tsetse fly. The single-celled parasite has a kinetoplast, which houses the cell's mitochondrial DNA, and a protruding flagellum that is crucial for cell movement. T. brucei undergoes major changes in shape and form during its developmental cycle. In one phase, known as the trypomastigote stage, the kinetoplast is located posterior to the nucleus and almost all of the flagellum is connected to the cell. In the epimastigote stage, on the other hand, the kinetoplast is anterior to the nucleus, and only part of the flagellum is fastened to the cell. T. brucei's close relatives come in many different shapes, indicating that the parasites have also altered their morphology during evolution.
When researchers from the University of Oxford reduced the expression of a protein called ClpGM6 in T. brucei trypomastigotes, the cells switched to an epimastigote-like morphology. The kinetoplast was close to the nucleus or anterior to it, and a long section of the flagellum extended beyond the cell. The parasites weren't identical to epimastigotes—they lacked a distinctive surface protein found at this life stage—but they were able to survive and reproduce for more than 40 generations.
ClpGM6 resides in the flagellar attachment zone and likely helps fasten the flagellum to the cell body. Loss of ClpGM6 shortened the flagellar attachment zone, which helps determine cell size and shape. The study suggests that dramatic morphological changes during the life cycle and during parasite evolution may result from adjustments in the levels of a few key proteins, rather than from wholesale changes in the parasite's protein or DNA content.
More information: Hayes, P., et al. 2014. J. Cell Biol. doi:10.1083/jcb.201312067
Journal information: Journal of Cell Biology
Provided by Rockefeller University