Nanoparticles with protein 'passports' evade immune system, deliver more medication to tumors
Scientists have found a way to sneak nanoparticles carrying tumor-fighting drugs past cells of the immune system, which would normally engulf the particles, preventing them from reaching their target. The technique takes advantage of the fact that all cells in the human body display a protein on their membranes that functions as a specific 'passport' in instructing immune cells not to attack them. By attaching a small piece of this protein to nanoparticles, scientists were able to fool immune cells in mice into recognizing the particles as 'self' rather than foreign, thereby increasing the amount of medication delivered to tumors.
Cancer Nanotechnology
Current approaches to chemotherapy leave patients with severe side effects because anti-cancer drugs meant to destroy tumors inadvertently kill healthy cells in the body. But scientists have recently developed nanoparticles that can ferry toxic medications directly to tumors while sparing healthy tissue. Because of their small size, nanoparticles escape from leaky blood vessels that are characteristic of tumors and accumulate in the cancerous tissue. Tumor cells take up the particles which release their toxic contents once inside. This localized delivery system allows doctors to give patients higher doses of medications than would normally be tolerated.
Previous attempts have been made to ward off attack by the immune system by coating nanoparticles densely with polyethylene glycol (PEG) "brushes" that physically block the adhesion of proteins that normally deposit onto foreign bodies to attract macrophages. While these brushes delay the onset of the immune response, they don't prevent it.
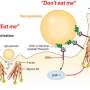
The inspiration for Discher's breakthrough work dates back thirteen years when a group of researchers showed with genetically engineered mice that a protein called CD47—which is found in the cell membranes of nearly all mammals—interacts with a receptor on macrophages called SIRPa, and, in doing so, signals that the cell is native and shouldn't be destroyed. The findings hinged on deleting mouse CD47 and raised many questions, including how such mice survive and whether there was relevance to humans.
Discher, who was engineering nanoparticles that self-assemble into various shapes at the time of the discovery, realized that the CD47-SIRPa mechanism for self-recognition could, in principle, be exploited to help nanoparticles sneak past the immune system. But it was also clear that human versions of purified proteins needed to be studied for any translation to humans.
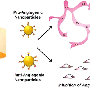
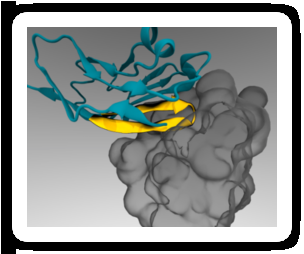
In 2008, Discher's lab demonstrated that human CD47 acts similarly to mouse CD47 as a "marker of self" via signaling through the SIRPa receptor. Shortly thereafter, a group of researchers elucidated the combined structure of human CD47 and SIRPa in atomic detail. Discher's lab used this information to conduct computer simulations and identify the smallest portion of CD47 that could still bind to SIRPa. The result was a short peptide that Discher's lab chemically synthesized and attached to standard nanoparticles.
"Reducing CD47 to an essential peptide was a key step," said Discher. "Sequencing of thousands of human genomes around the world has recently revealed many variations in the sequences of CD47 and SIRPa. We needed to engineer a 'universal' peptide that could bind SIRPa and function in all humans despite these differences."
Stealth nanoparticles avoid immune response
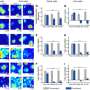
To test whether their peptide could help nanoparticles evade the immune system, Discher's team injected both peptide-bound nanoparticles and nanoparticles lacking CD47 into mice. Both types of nanoparticles contained a fluorescent dye that allowed the scientists to track the particles. In an article published on February 22, 2013 in Science, the researchers reported that in just thirty minutes post-injection of the particles, the mice's blood contained four times as many nanoparticles containing CD47 peptide as particles without the peptide, suggesting that CD47-bound particles were being viewed by macrophages as being similar to cells that belonged in the body.
Encouraged by these initial results, the team next filled their CD47-bound nanoparticles, as well as PEG-coated nanoparticles without CD47, with the anticancer drug paclitaxel plus a tumor-targeting antibody. The team separately injected both types of nanoparticles as well as Cremophore—the standard carrier for paclitaxel—into mice with human-like tumors. After just one day, the tumors in mice injected with CD47-bound nanoparticles were 70% the size of those injected with the PEG-coated nanoparticles. Additionally, CD47-bound nanoparticles were just as good or better at shrinking the tumors as Cremophore without causing any side effects. The team went on to document the molecular changes that occur inside macrophages when CD47 inhibits engulfment, suggesting additional medications might be used to inhibit clearance.
"Clinical trials using nanoparticles to deliver anticancer drugs are currently underway, but clearance by the immune system remains a significant hurdle," said Karen Peterson, Ph.D., Senior Advisor of Extramural Programs at NIBIB. "Discher's work is an elegant approach, which could enable other nanotherapeutics to be effective in clinical trials by providing a molecular "authentication" that the body does not recognize as foreign."
Peterson also noted the combination of bioengineering and computer modeling that went into generating the peptide; Discher's ability to test the function of differently sized peptides via computer simulation first, and then produce a man-made peptide based on these simulations allowed him to eliminate some of the guessing game, saving time and money in the long-run.
Future applications
Discher speculates that his CD47 peptide could be similarly used to prevent immune clearance of viruses used to deliver genes for gene-therapy treatment or to enhance biocompatibility and durability of larger foreign objects such as pacemakers and implants, whose parts can degrade over time due to attacks by the immune system.
Journal information: Science
Provided by National Institute of Biomedical Imaging and Bioengineering