May 17, 2011 feature
Smooth operators: Teflon microfluidic chips
(PhysOrg.com) -- The growing number of research and development efforts focused on microfluidics speaks to the technology’s promise of a potentially broad range of applications, largely in highly-integrated single-chip medical devices. However, the materials currently used to fabricate these labs-on-a-chip and other microfluidic devices have significant limitations, including absorption of small nonpolar and weakly polar molecules, adsorption of biomolecules, and the material’s molecules leaching into the microfluidic channel. The good news is that researchers have overcome these obstacles using microfluidic channels made entirely of Teflon, which supports cellular activity similar to that found in current materials. Moreover, whole-Teflon microchannels have gas permeability levels that permit cells to be cultured in-channel for extended periods of time.
The researchers, led by Prof. Hongkai Wu at Hong Kong University of Science and Technology’s Department of Chemistry, faced a number of obstacles to designing and developing a microfluidic chip that was optimally inert yet suitable to machining. “Currently, there are two major types of materials for microfluidic chips,” Wu explains to PhysOrg.com. “One is inorganic, such as glass and silicon. Unfortunately, fabrication of micropatterns and bonding chips of these materials are difficult and require sophisticated equipment. The other class of materials is plastics, including polydimethylsiloxane (PDMS) – the most widely-used – poly(methyl methacrylate) (PMMA), and polyurethane. Chips in plastics are easier to fabricate than in glass, but they have their own problems,” including the adsorption, absorption and leaching mentioned above, as well as being incompatible with organic solvents, all of which greatly limit their microfluidic chip applications.
“For example,” Wu continues, “they will be unsuitable for highly-sensitive analysis because the analyte will be lost by absorption if it’s a small, non/weakly polar molecule or by adsorption on channel walls if it’s a large molecule. For all of these reasons, we chose Teflon, which is well-known for its high degrees of inertness, non-adhesiveness and resistance to solvents.” Moreover, the Teflon compounds Wu used – perfluoroalkoxy (PFA) and fluorinated ethylenepropylene (FEP) – have melting points above 260 °C (one of the highest in thermoplastics) and are optically transparent (although less so than PDMS and glass).
At the same time, Teflon had its own challenges. For example, Wu notes, “Teflon’s superior inertness causes two major obstacles: one in micropatterning the material and the other in bonding patterned chips. Prior to our work, there were only several very expensive and complicated lithographic methods using high-energy radiation to effectively micropattern Teflon.”
In addition, Wu continues, “tight bonding of Teflon chips is rather difficult. Both bonding temperature and pressure need to be precisely controlled to overcome the problems that come from residue internal stress and plastic flow. “Initially we tried to bond the Teflon channel without pressure (as for bonding glass chips) and with constant pressure (as for bonding PMMA chips),” says Wu, “but neither worked. We needed an effective and convenient method to bond and thereby seal the Teflon channels.”
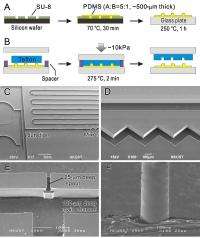
Overcoming these two obstacles led to the team’s two key innovations, micropatterning Teflon materials and bonding Teflon chips. “We established a very simple and easy-access method to fabricate three-dimensional Teflon micro- and nanostructures. The Teflon PFA and FEP substrates we used are melt-processable, so they could be hot-embossed using a template – an ideal way is to generate micropatterns in photoresist and then transfer the structure into the mold.”
But another problem arose. “Teflon’s melting points of substrates are much higher than those of photoresist. We therefore introduced a specially-treated intermediate thermosetting master to overcome the gap between low-melting-point photoresist master and a high-melting-point replica, allowing us to cast the master at milder temperatures into the replica and then use it at elevated temperatures to mold the patterns into Teflon.”
Their method was adapted from traditional soft-lithography, which previously wasn’t applicable to high-melting-point substrates. “Normal PDMS severely leaches gas above 150 °C, creating bubbles that make it impossible to mold micropatterns into Teflon. With our treated PDMS replica, we now can mold any micropatterns that are formed in photoresist by photolithography into thermoplastics (including Teflon) at as high as 350 °C.”
The team also designed a very simple and highly efficient method that solved the problems that have been encountered for a long time when bonding Teflon chips. This thermobonding process is based on different thermal expansion factors of Teflon materials (slightly higher than that of stainless steel) and the holding scaffold (stainless steel screw clamps) during bonding. “The bonding pressure is automatically controlled,” Wu explains. “When temperature is raised and the two Teflon plates are not bonded, Teflon expands more than the clamps and so the pressure is high. Once the two plates are bonded, the pressure is automatically released.”
Wu is already looking at future innovations and improvements. “We want to develop a smaller on-chip microvalve. The current nanoliter valve is still relatively large, so we’re working on reducing its volume to the picoliter range.” The team is also interested in advances in Teflon itself. “The optical transparency of our current whole-Teflon chip is still lower than that of PDMS and glass chips, so optical detection can be performed only when the optical path length of the Teflon chip is less than 2 mm. If the Teflon materials become as transparent as PDMS or glass, we will have more freedom in designing the microchips. But,” he acknowledges, “this depends on the design of new Teflon materials.”
In terms of the new chip’s most promising near-term and future applications, Wu comments that “since the whole-Teflon device is extremely inert and super-clean, it is superior for applications involving corrosive chemicals, strong solvents, high and low temperatures, and pressurized processes. It is antifouling and biocompatible, and therefore well suitable for quantitative and biological analysis. Interestingly, various biological cells can attach and grow well inside the Teflon channels – so it expands the applications of microfluidics to all these areas that were previously difficult. It is particular advantageous when accurate quantitative information is required.”
Beyond this Wu sees an even wider range of possibilities. “We also believe that Teflon materials are superior to PDMS for commercial applications of microfluidics due to their outstanding stability and reliability. Teflon could be a next-generation microchip material with very broad use, such as serving as standard equipment for flow reactors, microanalysis and bioassay. Moreover, they can also be used under extreme conditions – for example, on a space shuttle.”
Perhaps the ultimate application of Wu’s Teflon microfluidic technology will derive from the intersection of further miniaturization and biocompatibility. “We can mold Teflon pattern down to submicron range and fabricate sealed Teflon channels within the scale of 10 um. Since Teflon materials have outstanding biocompatibility and have long been used for implantations in human body, such as catheters, miniaturized Teflon microfluidic devices might be used for in-vivo diagnostics, drug delivery and flow control.”
More information:
* Whole-Teflon microfluidic chips, PNAS Published online before print May 2, 2011, DOI:10.1073/pnas.1100356108
* Department of Chemistry, Hong Kong University of Science and Technology
Copyright 2011 PhysOrg.com.
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of PhysOrg.com.