Holographic dark information energy

Holographic Dark Information Energy gets my vote for the best mix of arcane theoretical concepts expressed in the shortest number of words – and just to keep it interesting, it’s mostly about entropy.
The second law of thermodynamics requires that the entropy of a closed system cannot decrease. So drop a chunk of ice in a hot bath and the second law requires that the ice melts and the bath water cools – moving the system from a state of thermal disequilibrium (low entropy) towards a state of thermal equilibrium (high entropy). In an isolated system (or an isolated bath) this process can only move in one direction and is irreversible.
A similar idea exists within information theory. Landauer’s principle has it that any logically irreversible manipulation of information, such as erasing one bit of information, equates to an increase in entropy.
So for example, if you keep photocopying the photocopy you just made of an image, the information in that image degrades and is eventually lost. But Landauer’s principle has it that the information is not so much lost, as converted into energy that is dissipated away by the irreversible act of copying a copy.
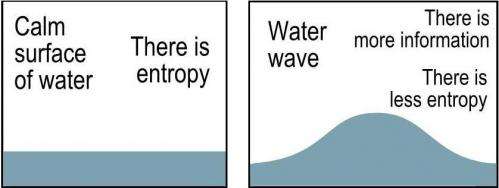
Translating this thinking into a cosmology, Gough proposes that as the universe expands and density declines, information-rich processes like star formation also decline. Or to put it in more conventional terms – as the universe expands, entropy increases since the energy density of the universe is being steadily dissipated across a greater volume. Also, there are less opportunities for gravity to generate low entropy processes like star formation.
So in an expanding universe there is a loss of information – and by Landauer’s principle this loss of information should release dissipated energy – and Gough claims that this dissipated energy accounts for the dark energy component of the current standard model of universe.
There are rational objections to this proposal. Landauer’s principle is really an expression of entropy in information systems – which can be mathematically modeled as though they were thermodynamic systems. It’s a bold claim to say this has a physical reality and a loss of information actually does release energy – and since Landauer’s principle expresses this as heat energy, wouldn’t it then be detectable (i.e. not dark)?
There is some experimental evidence of information loss releasing energy, but arguably it is just conversion of one form of energy to another – the information loss aspect of it just representing the transition from low to high entropy, as required by the second law of thermodynamics. Gough’s proposal requires that ‘new’ energy is introduced into the universe out of nowhere – although to be fair, that is pretty much what the current mainstream dark energy hypothesis requires as well.
Nonetheless, Gough alleges that the math of information energy does a much better job of accounting for dark energy than the traditional quantum vacuum energy hypothesis which predicts that there should be 120 orders of magnitude more dark energy in the universe than there apparently is.
Gough calculates that the information energy in the current era of the universe should be about 3 times its current mass-energy contents – which closely aligns with the current standard model of 74% dark energy + 26% everything else.
Invoking the holographic principle doesn’t add a lot to the physics of Gough’s argument – presumably it’s in there to make the math easier to manage by removing one dimension. The holographic principle has it that all the information about physical phenomena taking place within a 3D region of space can be contained on a 2D surface bounding that region of space. This, like information theory and entropy, is something that string theorists spend a lot of time grappling with – not that there’s anything wrong with that.
Source: Universe Today