Revealing a missing link
When it comes to genes, it’s definitely possible to have too much of a good thing. Accordingly, mammalian females have a mechanism that randomly inactivates one of the two X sex chromosomes within each somatic cell nucleus, ensuring that X-linked genes are represented to the same extent as in their single-X-bearing male counterparts.
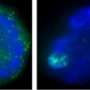
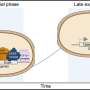
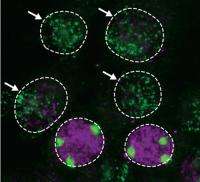
This process is executed by the product of the Xist gene. Although messenger RNAs typically get exported to the cytoplasm to produce protein, Xist RNA remains in the nucleus and accumulates along the surface of the X chromosome that is to be inactivated, and new findings from a team led by Shinichi Nakagawa at the RIKEN Advanced Science Institute in Wako have provided valuable insights into the mechanism behind this unusual localization.
Their screen of RNA-binding factors revealed a central role for heterogeneous ribonuclear protein U (hnRNP U) in regulating Xist distribution, and this RNA was scattered diffusely throughout the nuclei of cells in which hnRNP U levels were artificially reduced. Closer analysis indicated that hnRNP U acts as an intermediary that binds directly to both RNA and chromosomal DNA and tethers the two together. This physical association appears to be essential to X inactivation; although mouse embryonic stem cells lacking hnRNP U successfully initiated the maturation process, they were significantly more likely to exhibit gene activity from both X chromosomes.
Previous investigations have identified a structural role for hnRNP U within the nucleus, and at least one group has demonstrated that this protein tends to cluster near X chromosomes, although this potential aspect of its function remained unaddressed for the better part of decade. Indeed, Nakagawa was taken aback by its involvement in X inactivation. “I was surprised that we came across a factor that has been well-studied in the field of molecular biology rather than a ‘novel’ gene,” he says.
Although Xist is unique in its capacity to engineer the shutdown of an entire chromosome, there are numerous other non-protein-coding RNAs that contribute to the regulation of gene activity at a far smaller scale. Nakagawa hopes that this study will offer a window onto those mechanisms as well. “In most cases these non-coding RNAs control neighboring genes on the same chromosome, in a similar manner to Xist,” he says, “and it is possible that these non-coding RNAs are, in general, also retained around the site of transcription by hnRNP U.”
More information: Hasegawa, Y., et al.The matrix protine hnRNP U is required for chromosomal localization of Xist RNA. Developmental Cell 19, 469–476 (2010). Article
Provided by RIKEN