A miniature synchrotron for your home lab
In 2004 Lyncean Technologies announced the construction of the Compact Light Source (CLS), a miniature synchrotron which uses inverse Compton scattering to produce high-intensity, tunable, near-monochromatic x-ray beams. The CLS was designed to bring state-of-the-art protein structure determination to the home laboratory -- but it has also promised to have a broad impact across the spectrum of x-ray science.
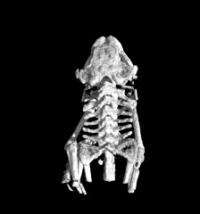
Today, at the 39th Winter Colloquium on the Physics of Quantum Electronics, Ronald Ruth, Ph.D., president of Lyncean Technologies, announced that the CLS has started delivering on this promise by achieving three key milestones using its unique x-ray beam: First scientific publication from the CLS featured by the Journal of Synchrotron Radiation on its January 2009 cover; first micro-tomographic images from the CLS; and first protein crystallography data set from the CLS.
The Compact Light Source prototype effort was funded by the National Institute of General Medical Sciences (NIGMS) Small Business Innovation Research (SBIR) program as an advanced technology related to the NIGMS Protein Structure Initiative (PSI). The CLS technology is based on an electron beam stored in a miniature storage ring colliding repeatedly with an opposing infrared light pulse stored in a high-finesse cavity. Each collision produces x-rays through inverse Compton scattering. The entire x-ray source fits in a 10x25 ft room similar in size to those used for Magnetic Resonance Imaging in medical clinics.
The first scientific publication using the CLS x-ray beam employs a technique called Differential Phase Contrast Imaging (DPCI) developed by Professor Franz Pfeiffer and collaborators at the Paul Scherrer Institute in Switzerland. DPCI uses a pair of micron-scale gratings to create two images, one sensitive to the phase of the x-ray wave front and the other sensitive to the local scattering power. This technique has been primarily developed at x-ray beam lines in large synchrotrons, and it relies on a small point-like x-ray source to achieve the coherence necessary for the fringes. The Compact Light Sources has an x-ray beam that is a perfect match for the technique.
Prof. Pfeiffer explained the motivation for imaging with the CLS x-ray beam, "High-resolution, soft-tissue imaging has been pursued at the large synchrotrons for the past 15 years, but to be useful to the biomedical community, the new methods must be able to use x-ray sources that are laboratory or clinical in scale. Differential Phase Contrast Imaging can use the full intensity of the CLS beam and can take advantage of its tiny source size and moderate divergence to image a field of view in the 10 cm range with very small pixel size. This will first open research and development applications such as small animal imaging, the study of tumor growth models or the development of Alzheimer's plaques in brain samples. Clinical applications might extend from mammography to osteoarthritis."
"We are only just beginning to exploit the benefits of the CLS for DPCI," continued Pfeiffer, "The JSR paper shows results from the very first experiments using the CLS for DPCI. Since then, we have also performed the first computed tomography using the CLS x-ray beam, and we are planning our next round of experiments this spring with higher x-ray energy (20 keV), higher intensity, and finer resolution."
In the second round of the Protein Structure Initiative, further CLS development was included in the Accelerated Technology Center for Gene to 3D Structure (ATCG3D, www.ATCG3D.org) supported by both NIGMS and the National Center for Research Resources (NCRR). "The focus of the CLS development has been towards protein crystallography," said Professor Ruth, "and with the ATCG3D Beta CLS we had the opportunity to improve performance and reliability. We now have the Beta CLS nearly ready for installation and further development."
"Using many of the ATCG3D CLS subsystems together with the prototype CLS", Ruth continued, "we saw our first protein diffraction in November and on our second crystal, we collected a data set with 3 Ĺ resolution. This was made possible by close collaboration with ATCG3D and in particular with Dr. Michael McCormick of the Peter Kuhn and Ray Stevens laboratory at The Scripps Research Institute. After the ATCG3D Beta CLS is commissioned, we will improve performance to make data collection rapid and routine."
Dr. Lance Stewart, director of ATCG3D and President of deCODE biostructures, commented on the imaging results and the first crystallography at the CLS, "The ATCG3D is very pleased to have had the opportunity to help develop the Compact Light Source. I believe the CLS technology will be important for both soft tissue imaging and protein crystallography."
"With the publication of the first scientific results and the collection of the first set of crystallography data using the CLS, the instrument has demonstrated its potential to have a broad impact on biomedical research," said Jeremy M. Berg, Ph.D., director of NIGMS. "The wide availability of an intense, tunable x-ray tool could transform numerous fields of research by improving access to a key resource."
On the web:
39th Winter Colloquium on the Physics of Quantum Electronics: www.pqeconference.com/pqe2009/
Journal of Synchrotron Radiation January cover: journals.iucr.org/s/issues/200 … /01/00/issconts.html
Lyncean Technologies: www.lynceantech.com
Source: Lyncean Technologies