What's really inside vapes? We pulled them apart to find out
While vapes or e-cigarettes first appeared around 20 years ago as an alternative to smoking, their prevalence and use have increasingly become problematic.

While vapes or e-cigarettes first appeared around 20 years ago as an alternative to smoking, their prevalence and use have increasingly become problematic.
Analytical Chemistry
Jun 27, 2024
0
85

The amount of dietary sugars and the administration of lithium both impact the lifespan of the fruit fly Drosophila melanogaster. It is noteworthy that lithium is attributed with insulin-like activity as it stimulates protein ...
Molecular & Computational biology
Jun 21, 2024
0
0

A new method in spectromicroscopy significantly improves the study of chemical reactions at the nanoscale, both on surfaces and inside layered materials. Scanning X-ray microscopy (SXM) at MAXYMUS beamline of BESSY II enables ...
Nanomaterials
Jun 17, 2024
0
15

Spontaneous parametric down-conversion (SPDC), as a source of entangled photons, is of great interest for quantum physics and quantum technology, but so far it could be only implemented in solids.
Optics & Photonics
Jun 13, 2024
0
43

As the electric vehicle market booms, the demand for lithium—the mineral required for lithium-ion batteries—has also soared. Global lithium production has more than tripled in the last decade. But current methods of extracting ...
Materials Science
Jun 8, 2024
0
293

Researchers have developed a safer, cheaper, better performing and more flexible battery option for wearable devices. A paper describing the "recipe" for their new battery type was published in the journal Nano Research Energy ...
Nanomaterials
Jun 5, 2024
0
0

Until now, the generation and storage of electricity from solar energy has been dependent on various devices, leading to conversion losses. That may change soon, as chemists at Friedrich-Alexander-Universität Erlangen-Nürnberg ...
Analytical Chemistry
Apr 19, 2024
0
29
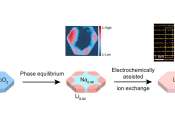
Ion exchange is a powerful technique for converting one material to another when synthesizing new products. In this process, scientists know what reactants lead to what products, but how the process works—the exact pathway ...
Analytical Chemistry
Apr 16, 2024
0
10

Chemists at the Department of Energy's Oak Ridge National Laboratory have invented a more efficient way to extract lithium from waste liquids leached from mining sites, oil fields, and used batteries. They demonstrated that ...
Analytical Chemistry
Apr 16, 2024
1
38

There's a reason airlines won't let you put your laptop in your checked luggage; the lithium-ion battery poses a serious fire hazard. But why? Lithium is incredibly reactive. For instance, pure lithium violently interacts ...
Earth Sciences
Apr 15, 2024
0
11
Lithium (pronounced /ˈlɪθiəm/) is the chemical element with atomic number 3, and is represented by the symbol Li. It is a soft alkali metal with a silver-white color. Under standard conditions it is the lightest metal and the least dense solid element. Like all alkali metals lithium is highly reactive, corroding quickly in moist air to form a black tarnish. For this reason lithium metal is typically stored under the cover of oil. When cut open lithium exhibits a metallic luster, but contact with oxygen quickly turns it back to a dull silvery gray color. Lithium in its elemental state is highly flammable.
According to theory, lithium was one of the few elements synthesized in the Big Bang. Since its current estimated abundance in the universe is vastly less than that predicted by theory; the processes by which new lithium is created and destroyed, and the true value of its abundance, continue to be active matters of study in astronomy. The nuclei of lithium are relatively fragile: the two stable lithium isotopes found in nature have lower binding energies per nucleon than any other stable compound nuclides, save for the exotic and rare deuterium, and 3He. Though very light in atomic weight, lithium is less common in the solar system than 25 of the first 32 chemical elements.
Due to its high reactivity it only appears naturally in the form of compounds. Lithium occurs in a number of pegmatitic minerals, but is also commonly obtained from brines and clays. On a commercial scale, lithium metal is isolated electrolytically from a mixture of lithium chloride and potassium chloride.
Trace amounts of lithium are present in the oceans and in some organisms, though the element serves no apparent vital biological function in humans. However, the lithium ion Li+ administered as any of several lithium salts has proved to be useful as a mood stabilizing drug due to neurological effects of the ion in the human body. Lithium and its compounds have several industrial applications, including heat-resistant glass and ceramics, high strength-to-weight alloys used in aircraft, and lithium batteries. Lithium also has important links to nuclear physics. The transmutation of lithium atoms to tritium was the first man-made form of a nuclear fusion reaction, and lithium deuteride serves as a fusion fuel in staged thermonuclear weapons.
This text uses material from Wikipedia, licensed under CC BY-SA