October 25, 2023 report
This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Experiments show molecules, not substrates, are mostly responsible for chirality-induced spin selectivity
A team of chemists at Northwestern University, working with a pair of colleagues from Università di Parma, reports that the chirality-induced spin selectivity (CISS) effect is caused by the molecules involved, not a substrate, when quantum spin is transmitted from one electron to another while moving through a material.
In their paper published in the journal Science, the group describes their experiments and possible uses for the results. Joseph Subotnik, a chemistry professor at the University of Pennsylvania, has published a Perspective piece in the same journal issue outlining the work done by the team on this effort.
As Subotnik notes, learning about the ways that electrons make their way through molecules and the means by which they carry energy with them plays a critical role in the development and manufacture of many types of modern technologies, from light-emitting diodes to photovoltaic cells. As part of that effort, scientists have discovered what is now known as the CISS effect, where the left-or right-handedness of a molecule can affect how electrons with different spins move through a material.
Prior research involving attempts to observe the CISS effect in action have all involved the use of a substrate, suggesting that one is needed for the effect to happen. In this new effort, the researchers found a way to observe the CISS effect in isolated molecules, proving that it is the molecules that are responsible for the phenomenon.
To observe the effect in isolation, the team started by creating molecules with three main parts: an electron donor, a bridge and an electron acceptor. They then grew crystals made from the molecules and aligned them using a magnetic field and then froze them in place. In such a configuration, it was possible for the researchers to change the chirality of the crystal by spinning the apparatus holding them.
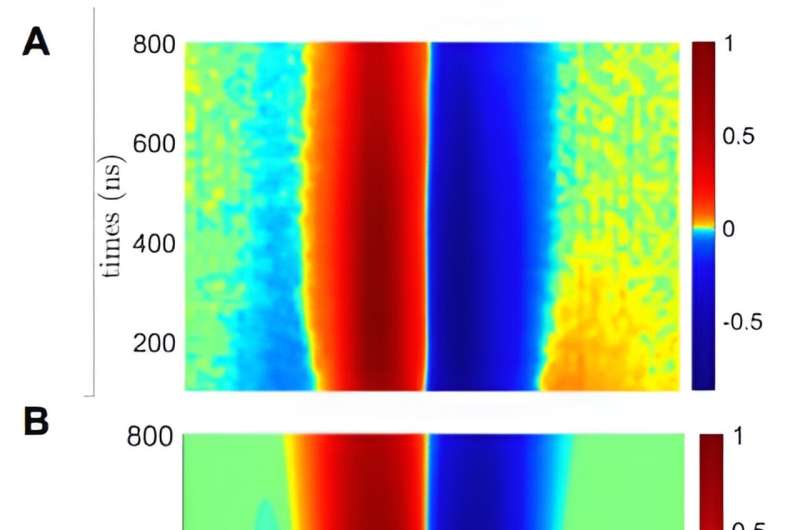
To observe the CISS effect, the team fired a laser at the donor molecule, forcing the release of an electron, which traveled over a bridge and joined the receiver molecule. The team monitored the trip using electron paramagnetic resonance spectroscopy—the measurements showed that the traveling electron's spin was impacted by the chirality of the bridge molecule. That proved that a substrate was not needed.
More information: Hannah J. Eckvahl et al, Direct observation of chirality-induced spin selectivity in electron donor–acceptor molecules, Science (2023). DOI: 10.1126/science.adj5328
Joseph E. Subotnik, Chiral molecules to transmit electron spin, Science (2023). DOI: 10.1126/science.adk5634
Journal information: Science
© 2023 Science X Network