This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Hot electron electrochemistry with ultrafast laser pulses
Laser-induced electrochemical deposition of metals on metals relies possibly on thermal and defect generation effects. When semiconductor substrates are chosen, locally photogenerated electrons can reduce metal ions resulting in metallic surface structures. Laser-induced electrochemical de- and repassivation investigations can support in-situ corrosion research studies.
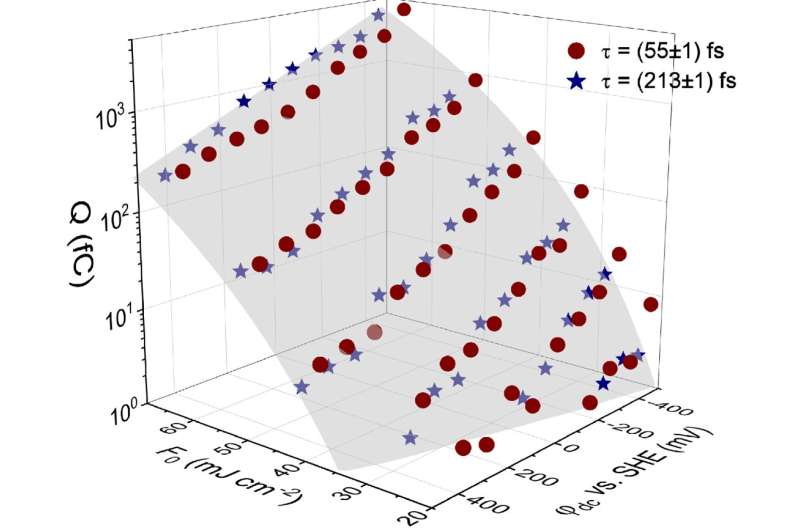
The thermoemission of nonequilibrium electrons was identified as a unique technique for the generation of picosecond current pulses which cannot be realized by conventional instrumentation. This allows short-lived intermediates of electrode reactions to be studied. Barring kinetic hindrance demanding the application of a certain overpotential, electrochemical reactions occur once the energy levels of the redox active species present in the electrolyte are crossed by the Fermi level of the electrode. Femtosecond laser pulse induced hot electron electrochemistry in aqueous solution allows a new perspective in the investigation of the electrode/electrolyte interface, the reactional pathways taken by short-lived intermediate species and laser processing in solution as a whole.
The term femtochemistry dates back to the pioneering work of Zewail, who employed femtosecond laser pulses in the investigation of molecular dynamics. Since molecular motion as well as chemical bond breaking and forming occurs on a femtosecond timescale, pump-probe experiments with femtosecond laser pulses allowed the first real time observation of the transition states of model chemical reactions. In 1999 Zewail was awarded the Nobel Prize in Chemistry for his work on the disintegrations of iodocyanide ICN and sodium iodide NaI. Surface femtochemistry, concerning the time resolved observation of reactions of adsorbates at various interfaces, oftentimes relies upon hot electron induced processes. Those hot carriers are emitted from metals and semiconductors under experimental conditions.
In the past, hot carrier injection (HCI) was mainly regarded as an undesirable effect in device engineering. In fact, HCI is a main reason for transistor aging, with hot electrons or holes (depending on the doping of the transistor) tunneling into the gate dielectric. This leads to the build-up of charge within the dielectric layer, increasing the switching threshold voltage. As a result, the switching time of the device increases. Recently, however, the possibility to capitalize on this non-equilibrium effect has drawn active interest from the scientific community.
The desorption and reaction behavior of co-adsorbed O and CO on a Ru(0001) surface are a prime example for the profound insight hot electron driven femtochemistry can give into molecular dynamics at surfaces. Whereas the heating of the surface leads exclusively to CO desorption, the treatment with 100 fs near-infrared laser pulses (800 nm) opens a new reaction pathway. The non-equilibrium emission of hot electrons excited by the ultrashort laser pulses leads to the oxidation of CO to CO2. Double pulse experiments revealed the electron-mediation of the oxidation as opposed to the phonon-mediation of the CO desorption. The Ru-O bond activation was identified as the rate determining step.
The advent of hot carrier mediated energy transfer has also reached photovoltaics and photocatalysis. The novel working principle is the plasmonically driven direct conversion of (solar) light into electric energy in metal nanostructures. A common hot electron harvesting method is the transfer through a Schottky junction into the conduction band of a contacting semiconductor. An exemplary application of hot carriers in the optimization of the conversion efficiency of photovoltaic and photocatalytic devices is, e.g., the pairing of conventional semiconductor absorbers with metallic nanoparticles, resulting in a new energy-harvesting pathway. Light with a wavelength too long to be absorbed by the semiconductor can excite plasmon-enhanced hot electron emission in the neighboring metal. The emitted hot electrons are in turn able to pass into the conduction band of the semiconductor.
Moreover, the dissociation of H2 at room temperature induced by the hot electron transfer from gold nanoparticles to adsorbed gas molecules is an intriguing process. Visible light at plasmon resonances brings the gold electron system out of equilibrium. Under the chosen experimental conditions, the excited plasmons decay nonradiatively, raising electrons to transient energy levels below vacuum. Due to their higher energy, the hot electrons are able to trespass the electronic equilibrium distribution and transfer into an anti-bonding state of a close hydrogen molecule.
A practical implementation is autonomous water-splitting. It is based on an array of gold nanorods capped with a TiO2 layer to form a Schottky junction. The TiO2 layer is coated with platinum nanoparticles. The excitation process of the hot electrons is again plasmonically driven to increase efficiency. Excited hot electrons reach the gold nanorod surface and pass into the conduction band of the semiconductor layer. From there, they are captured by the nearby platinum nanoparticles, which in turn catalyze the hydrogen ion reduction. The residual defect electrons in the gold nanorod are replenished by a cobalt-based oxygen evolution catalyst also situated on the rod. Also hot electrons may be used to charge platinum nanoparticles, initiating chemical reactions. A similar approach with a broader perspective on electrochemical analysis was the voltage induced injection of hot electrons into the silver top layer of a metal-insulator-metal element, which allows the investigation of electrochemical processes occurring at significantly different potentials as in the equilibrium case.
In the group of Prof. Kautek, sub-picosecond laser pulses were employed to study the hot electron emission from metal electrodes into electrolyte solution. This elegant approach got rid of a pre-fabricated junction as an electron filter and relied purely on the properties of the electrochemical double layer. This way, the hot electrons were directly injected into the electrolyte solution, allowing the study of the metal/electrolyte interface and short-lived intermediates formed close to the electrode. The work is published in the journal Opto-Electronic Advances.
More information: Oskar Armbruster et al, Hot electron electrochemistry at silver activated by femtosecond laser pulses, Opto-Electronic Advances (2023). DOI: 10.29026/oea.2023.220170
Provided by Compuscript Ltd