Crystal phase engineering offers glimpse of future potential, researchers say
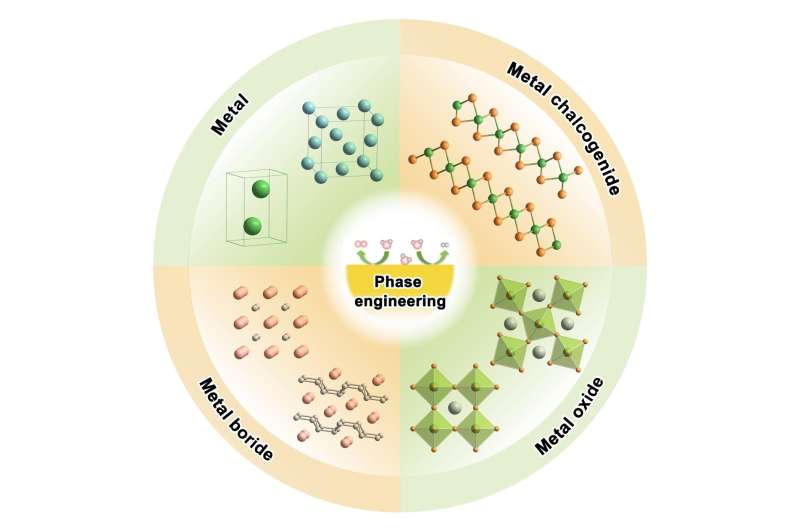
Atomic rearrangement changes a material's physical and chemical properties, which may lead to potential applications across disciplines, including in sustainable energy. With two decades of focused attention on how regulating such rearrangements—a process called phase engineering—may enable sustainable energy conversion processes, researchers in China have summarized the work so far, including how the field might progress.
They published their review on July 11 in Nano Research, with a specific focus on electrocatalysts. These materials trigger, enhance or resolve the chemical and electrical reactions involved in converting energy into storable or usable formats. They often serve as an electrode or as an electrode component.
"Phase engineering is an important strategy for designing efficient electrocatalysts toward these energy conversions, because it enables all catalytically active atoms to rearrange and form new lattices," said co-corresponding author Xiaoxin Zou, professor, State Key Laboratory of Inorganic Synthesis and Preparative Chemistry, College of Chemistry, Jilin University. "This provides great opportunity to rationally manipulate atoms to discover attractive structural frameworks and to achieve better electrocatalysis. And while, in recent years, several researchers have summarized the preparation of nanomaterials with novel arrangements, this is the first systematic review toward rationalizing how these phases influence electrocatalytic activity."
These various atomic arrangements are known as crystal phases. By changing how the atoms are arranged on the surface of a solid material, or in its bulk, can drastically change what the material can do. Zou noted, however, that the surface is essentially an extension of the bulk and cannot exist independently, so their connection is key to developing desirable and stable electrocatalysts.
"The underlying logic of phase engineering lies in an intimate relationship between the properties of the surface and of the bulk of a catalyst," Zou said. "Engineering the bulk phase of a catalyst, which directly influences the surface, is a powerful strategy to design smart catalysts both internally and externally."
The crystal structure of the bulk determines the material's electronic structure, its conductivity and, largely, the composition of the surface layer. Different bulk crystal structures possess different characteristics and surface energies, leading to diverse morphology and catalytically active sites. Even for catalysts that experience significant surface damage or reconstruction during the catalysis process, Zou said, the bulk's initial crystal structure strongly influences reconstitution and the final structure of the surface.
Over the last 20 years, several researchers have investigated this relationship, exploring unconventional electrocatalytic phases and how to induce such transformations. Driven by the demand for sustainable energy conversion processes, such as nitrogen fixation and carbon dioxide reduction, researchers advanced characterization techniques, as well as the theory underlying experimental work.
"These things made it possible to precisely and accurately understand the effects of crystal phases on electrocatalytic performance," Zou said. "So, it is time to summarize phase engineering-related research that helps unravel phase-performance relationships and refines prediction in electrocatalysis studies."
Next, Zou and his team recommend that researchers pursue four main areas to further advance crystal phase engineering for catalysis research.
"To develop competent catalysts for different energy conversion processes from a phase focus, we propose exploring the relationship between the crystal phase and catalytic activity levels; combining phase engineering with other design strategies; unraveling the formation and evolution mechanisms of unconventional phases; and enriching catalytic research of more fluid phases," Zou said.
More information: Hui Chen et al, Crystal phase engineering of electrocatalysts for energy conversions, Nano Research (2022). DOI: 10.1007/s12274-022-4605-y
Journal information: Nano Research
Provided by Tsinghua University Press