Confocal laser speckle autocorrelation imaging of dynamic flow in microvasculature
In a new publication from Opto-Electronic Advances, researchers from the National University of Singapore, Singapore, discuss confocal laser speckle autocorrelation imaging of dynamic flow in microvasculature.
Quantitative flow measurement and visualization is vital for many scientific and engineering disciplines. The authors of this paper propose a label-free dynamic flow imaging method, confocal laser speckle imaging, for real-time and quantitative imaging of blood flow on the microscopic level. The imaging system developed shares many features of a confocal fluorescence microscope and is, therefore, able to obtain high-quality, detailed flow images from thick tissue samples. The method described here does not require fluorescence labeling or any other sample preparation procedure.
Instead, the contrast mechanism is purely intrinsic and based on optical phase changes caused by flowing blood cells, which can be converted into random light intensity fluctuations. When a tissue sample is illuminated with a laser beam, the acquired images generally contain such random intensity fluctuations, the so-called laser speckles. The confocal laser speckle imaging setup is implemented on top of a line-scan confocal microscope, which forms an illumination line on the sample. A line camera is positioned to selectively capture the speckle signals coming from the illuminated line and effectively reject the out-of-focus light, which is a serious problem leading to reduced contrast and resolution in conventional laser speckle imaging techniques.
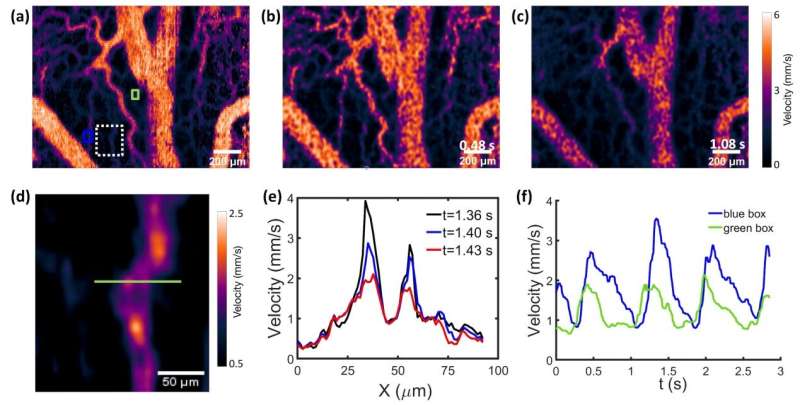
By quickly scanning the illumination line across the sample surface, two-dimensional raw speckle images can be acquired at a speed of greater than 200 frames per second. Time series analysis of the speckle images is performed pixel by pixel, a strategy that preserves the spatial resolution in the processed images. Autocorrelation and speckle contrast calculation are both commonly used analysis methods that link the speckle derived parameters to the local blood flow velocity. However, the combination of confocal microscopy with autocorrelation based speckle analysis, which is called Line Scan Laser Speckle Autocorrelation Imaging (LSAI), proves to be superior.
With small animal imaging experiments, the authors demonstrated that LSAI is able to quantify the local flow velocity at individual pixels, which are significantly smaller than the typical diameter of capillaries. Moreover, LSAI is fast enough to capture video-rate flow velocity changes at the same microscopic level. In short, confocal laser speckle imaging brings a breakthrough to in vivo flow imaging with its unprecedented performance.
An immediate application of confocal laser speckling imaging is to map and quantify dynamic blood flow in microvessels. Microvessels are the smallest blood vessels within organ tissues, including terminal arterioles, metarterioles, capillaries, and venules. Inside the microvessels network, the interaction between blood and tissue creates an environment for tissue cells to survive. The circulation of the blood in the microvasculature is so-called microcirculation, which is fundamental for analyzing and understanding the pathophysiology and pathogenesis of a wide range of human diseases. Experimental tools with adequately high temporal resolution and spatial resolution are highly desirable for in vivo visualization, and more importantly, quantitative measurement of the time-dependent blood flow maps in the microvasculature for further clinical and preclinical investigations. The novel confocal laser speckle imaging method developed by the authors of this article overcomes the technical limitations of existing techniques. It may become a standard imaging tool in microcirculation research as well as clinical diagnoses.
More information: Du E, Shen SH, Qiu AQ, Chen NG, Confocal laser speckle autocorrelation imaging of dynamic flow in microvasculature, Opto-Electronic Advances (2022). DOI: 10.29026/oea.2022.210045
Provided by Compuscript Ltd