February 25, 2022 feature
3D micromesh-based hybrid printing for microtissue engineering
Bioprinting is widely applicable to develop tissue engineering scaffolds and form tissue models in the lab. Materials scientists use this method to construct complex 3D structures based on different polymers and hydrogels; however, relatively low resolution and long fabrication times can result in limited procedures for cell-based applications.
In a new report now available in Nature Asia Materials, Byungjun Lee and a team of scientists in mechanical engineering at Seoul National University, Seoul, Korea, presented a 3D hybrid-micromesh assisted bioprinting method (Hy-MAP) to combine digital light projection, 3D printed micromesh scaffold sutures, together with sequential hydrogel patterning. The new method of bioprinting offered rapid cell co-culture via several methods including injection, dipping and draining. The work can promote the construction of mesoscale complex 3D hydrogel structures across 2D microfluidic channels to 3D channel networks.
Lee et al. established the design rules for Hy-MAP printing via analytical and experimental investigations. The new method can provide an alternative technique to develop mesoscale implantable tissue engineering constructs for organ-on-a-chip applications.
Materials engineering: Developing 3D micro-mesh platforms (3D MMP)
Using soft lithography-based microfluidic platforms, bioengineers can pattern multiple cell types for tissue engineering and organ-on-a-chip instruments. The concept of capillary burst valve has led to active research on microfluidic cell culture platforms via a phenomenon known as liquid pinning, where liquid can be maintained in the microstructure through capillary burst pressure (CBP) to form a predesigned shape of liquid. The method of liquid patterning by capillary burst valve (CBV) is explored via surface hydrophilicity modification, to develop micropost structures in microfluidic channels to hold liquid and hydrogel cells, for barrier-free co-culture. Since the devices are two-dimensional (2D) and difficult to develop via conventional soft lithography, scientists have incorporated patterning cells in 3D via 3D printing methods.
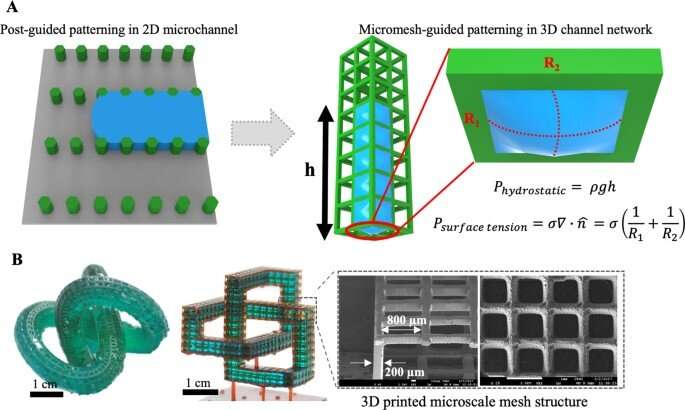
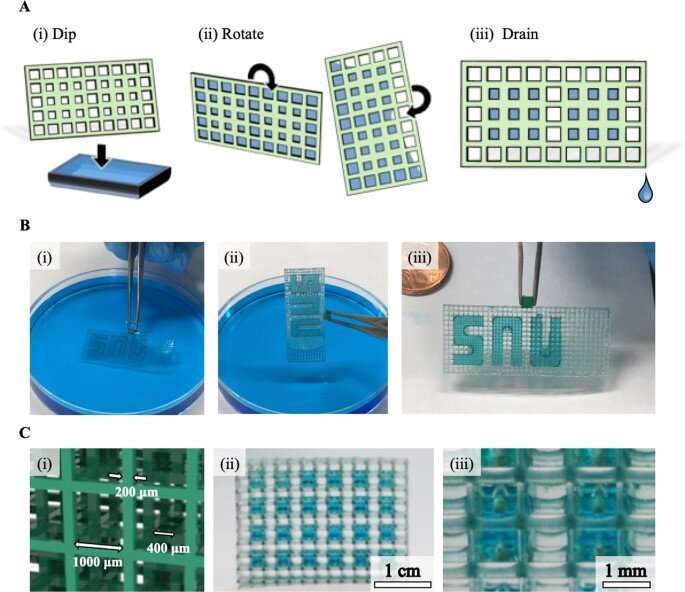
In this work, Lee et al. formed a new concept of hybrid-micromesh assisted bioprinting (Hy-MAP) to combine high-resolution 3D printed micromesh structures with capillary burst, valve-based rapid hydrogel patterning to form a 3D micro-mesh platform (3D-MMP) containing liquid in designed areas, through high-resolution digital light-projection printing. They also developed an additional method to inject liquid into a simple channel to pattern hydrogels and cells by incorporating a mesh framework to contain liquid based on surface tension. The team showed the advantage of open microfluidics and proposed a method to bioengineer 3D blood vessel networks by patterning hydrogels and cells on a 3D Micro-Mesh platform with hybrid micromesh-assisted printing (Hy-MAP) for tissue engineering applications.
Constraining liquid within the 3D micromesh structure
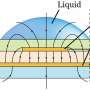
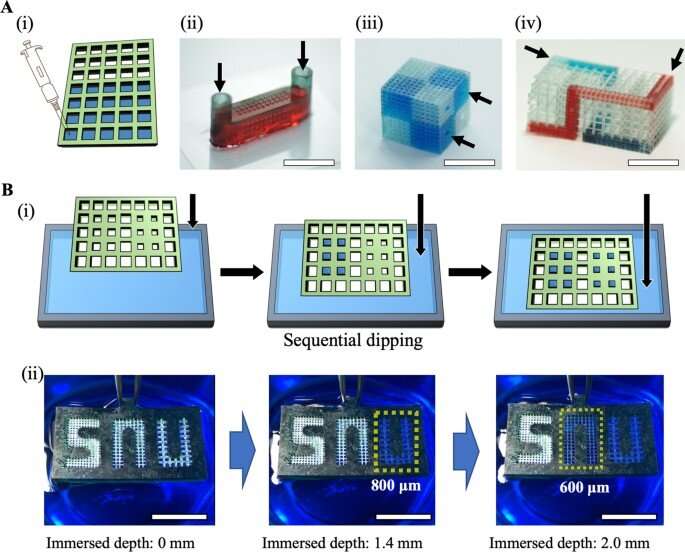
Since the surface tension between the liquid and solid surface of the microstructures can facilitate liquid shape maintenance below a specific pressure limit, Lee et al. developed the micromesh structure to regulate the behavior of microliquids or droplets within a 3D channel. Lee et al. used photocurable polymer resin to construct the micromesh; the resulting 3D MMP (Micro-Mesh Platform) had a thickness of 200 µm and the liquid within maintained an outwardly convex appearance due to surface tension.
The team obtained images of the micromesh via scanning electron microscopy to highlight a material with high accuracy, without defects. As a result of 3D printing and computer-aided technology, the research team formed complex 3D structures that were not hitherto obtainable through conventional manufacturing methods, including soft lithography and injection molding.
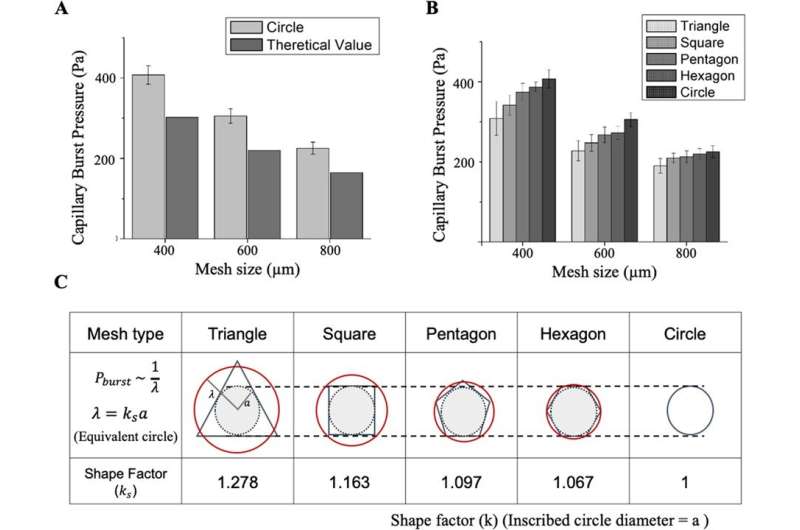
Theory and experiments with capillary burst pressure (CBP)
In order to fill the liquid in designated positions, the team showed how the pressure applied to the micromesh had to be below the capillary burst pressure (CBP), where the CBP increased as the size of the mesh decreased. Regardless of the flawlessly accurate 3D printing method, the micromesh lacked sharpness during the process of printing, which caused deviations between the observed experimental and known theoretical values. The team conducted several analyses to show how CBP varied the size of the gap while retaining water within simple micromesh units of different shapes and sizes. Based on these principles, Lee et al. produced a wide range of microfluidic channels. The Hy-MAP (hybrid micromesh-assisted printing) method was also advantageous. The team showed how constructs could be manufactured for selective liquid patterning to form a complex 3D channel.
Cell patterning for tissue engineering applications
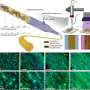
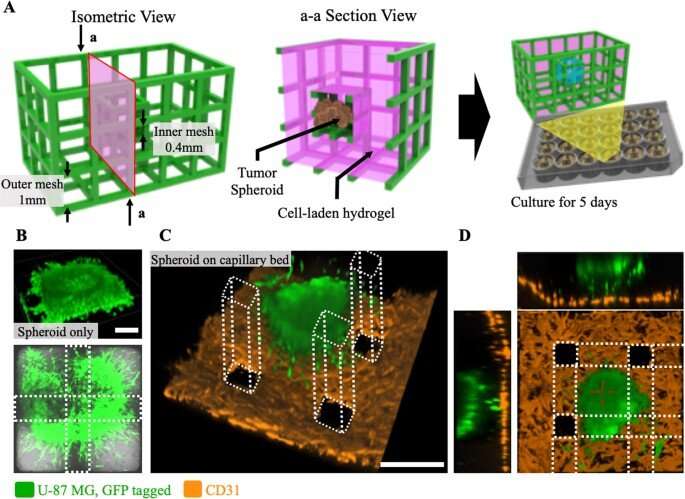
The scientists developed a range of 3D patterning methods to contain fluids and hydrogel encapsulated cells. By using the Hy-MAP method they introduced sophisticated cell patterning as a potential alternative to 3D bioprinting. To complete the cell culture, Lee et al. formed a compartmentalized structure for selective patterning of multiple cell types. They then prepared a mixture of cells and hydrogels prior to enzymatic gelation of the culture setup and conducted many co-culture studies with endothelial cells, stromal cells and cancer cells.
During the experiments, they injected a cell-laden fibrin gel into a three-channel micromesh platform, within which they cultured a 3D vasculature to facilitate vasculogenesis. The team similarly constructed a micromesh platform for tumor-vasculature coculture experiments, and based on the studies, they showed how cells could be cultured across several methods, to effectively integrate Hy-MAP for tissue engineering.
Outlook
In this way, Byungjun Lee and colleagues presented a new hybrid bioprinting system with a 3D printer and patterning method to apply fluid flow in fluid mechanics and pattern fluids at the microscale in cell culture platforms. The team overcame limits of conventional bioprinting systems by filling a liquid in a simple channel or a channel of any other conformation to confine liquids within a mesh framework via surface tension to generate liquid or hydrogel patterns. This setup is effective across diverse 3D organized, multipart cell culture platforms for cell biology, drug-efficiency and drug toxicity tests and is suited for advanced lab-on-a-chip and organ-on-a-chip applications, together with biodegradable micromesh materials developed for applications in regenerative tissue engineering .
More information: Byungjun Lee et al, 3D micromesh-based hybrid bioprinting: multidimensional liquid patterning for 3D microtissue engineering, NPG Asia Materials (2022). DOI: 10.1038/s41427-022-00355-x
Bagrat Grigoryan et al, Multivascular networks and functional intravascular topologies within biocompatible hydrogels, Science (2019). DOI: 10.1126/science.aav9750
Journal information: Science
© 2022 Science X Network