Stable armchairlike hexazine ring in tungsten hexanitride
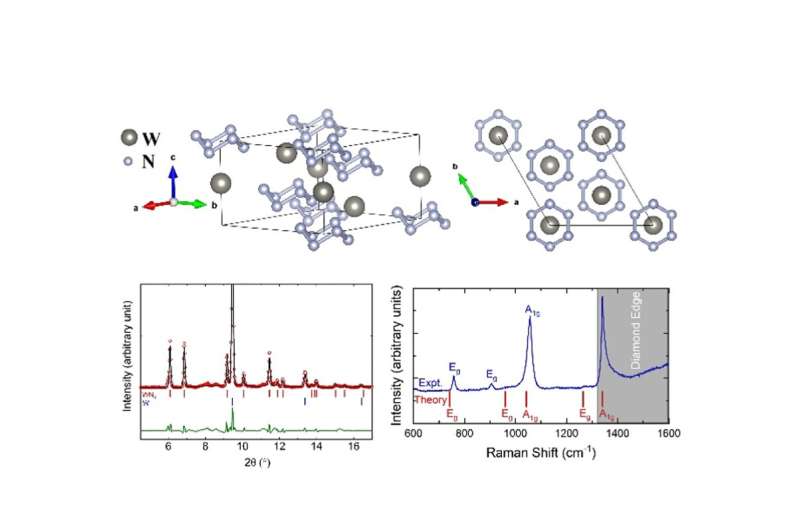
Tungsten hexanitride with armchairlike hexazine N6 ring has been synthesized by a group of scientists led by Dr. Jin Liu and his former postdoc Nilesh Salke at HPSTAR (Center for High Pressure Science & Technology Advanced Research). WN6 is a promising high-energy-density and super-hard material. Their findings are published in the recent issue of Physical Review Letters.
Diatomic nitrogen is the most abundant molecule in Earth's atmosphere accounting for almost 78% volume. The strong triple bond in nitrogen makes it very stable and unreactive at near ambient conditions. However, under intense pressure and high-temperature conditions, nitrogen will behave entirely differently, it can form double- or even single-bonded structure or react with other elements to form novel nitrides. Single-bonded polymeric nitrogen or nitrides possessing single-bonded nitrogen are of great scientific interest as a high-energy-density material. And transition metal nitrides are the very promising candidates that might contain the planar nitrogen hexazine (N6) ring which are predicted to be impossible to stabilize experimentally due to the lone pair repulsion.
The team created WN6 in a laser-heated diamond anvil cell by elemental reaction between tungsten and nitrogen above pressure of about 1.3 Mbar and temperature of ~3500 K. In-situ synchrotron X-ray diffraction (XRD) allowed them to identify the tungsten hexanitride phase, crystallizing with novel armchair-like N6 rings, and the high-pressure Raman spectroscopy measurement confirmed the presence of N-N single bonds in N6 rings. Further theoretical calculations also support their experimental observations.
"The armchair-like hexazine nitrogen sublattice in the WN6 is remarkable and comparable to that in the polymeric nitrogen phases, making it a promising high-energy-density material candidate," said Dr. Nilesh Salke, now a postdoctoral researcher at the University of Illinois at Chicago.
Additionally, WN6 shows a Vickers hardness of up to 57 GPa, the highest hardness among all transition metal nitrides along with good toughness. They credited the ultra-stiffness of WN6 to balance between the attractive interaction of N6 rings with W atoms and the repulsive interaction of N6 rings with each other based on theoretical calculations.
"To our knowledge, this is the first experimental report on the single-bonded transition metal nitride," said Dr. Jin Liu, "We believe that this work will stimulate further experimental efforts to synthesize other nitrides with novel structural, chemical, and physical properties."
"Our experimental demonstration of stabilizing armchairlike hexazine N6 ring in WN6 paves the way for future efforts to stabilize planar hexazine ring," added Dr. Liu.
More information: Nilesh P. Salke et al, Tungsten Hexanitride with Single-Bonded Armchairlike Hexazine Structure at High Pressure, Physical Review Letters (2021). DOI: 10.1103/PhysRevLett.126.065702
Journal information: Physical Review Letters
Provided by Center for High Pressure Science & Technology Advanced Research