Some surprises about the degradation of microplastics in our oceans

Not all the news about plastic in the ocean is what we expect. In fact it may be not quite as bad as initially thought. This comes as welcome information as we are celebrating National Science Week with an oceans theme.
Recently research led by an ANSTO scientist has found that the structural degradation of plastic in the ocean facilitates its entry into the natural carbon cycle efficiently as carbon dioxide.
The research was an investigation of the fragmentation of packaging into microplastics in the ocean.
The work does not diminish the serious threat to wildlife from large pieces of packaging but draws important conclusions to the factors determining the lifetime of plastics in the environment.
The study was led by Dr. Chris Garvey (Instrument Scientist at ANSTO's Australian Center for Neutron Scattering). Chris is currently a Fellow of the Lund Institute of Advanced Neutron and X-ray Science while based at the University of Malmö in Sweden.
The work brings important understanding of the molecular scale physical mechanisms which enable the fragmentation of plastics into the ocean.
"Cellulose waste, including cardboard and paper, enters the carbon cycle by a well understood process. In recent years plastics, and in particular polyethylene, which originate from fossil fuels, have replaced paper as a barrier material for packaging. It is important to understand how this carbon, from a fossil pool, enters the carbon cycle," said Garvey.
Obviously with exposure to UV sunlight and oxygen in the ocean plastic begins to get brittle, crack and break into smaller pieces.
Garvey and his associates wanted to know the molecular scale process leading to embrittlement and if these processes slow or hasten the chemical degradation of plastics.
The microplastics used in the experiments included samples that were collected in the tropical waters of the Caribbean as part of the Atlantic gyre.
These samples were compared slightly larger weathered pieces of plastic from the same source and new samples that were the matching source of the weathered pieces.
The microplastics, which were about a millimeter in size, had been in the water a long time but there is no way of knowing when they entered the ocean other than they represent significant fragmentation of the original packaging.
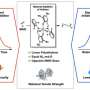
However, probes with analytical techniques, most specially small and wide X-ray and Raman scattering, identified important changes to the microstructure.
Plastics, in this example, polyethylene, consist of extremely long molecules which span many layers of alternating layers of crystalline polymer chains forming a lamella structure.
This is the normal structure of polyethylene produced by injection molding which is used for packaging.
The natural tendency of the long chain molecules is to crystallize and this process is frustrated by the entanglement of the polymer chains between the crystalline lamellae.
UV radiation in sunlight causes the chains to be cut. This has implications for the major degradation pathway which ultimately converts the polymer chains into carbon dioxide.
"This releases the kinetic arrest, so the polymer starts to crystallize slowly again and this crystallization process disrupts the lamella structure," explained Garvey.
"When the lamella is disrupted, it is no longer such an effective barrier and oxygen can diffuse more easily into it," he said.
In the study of polyethylene from different packaging, wide-angle X-ray scattering provided an indication of the nanoscale fragmentation of the crystalline layers and an increase in the fraction of crystalline polymer.
Small-angle X-ray scattering demonstrated loss of the alternating layers of amorphous and crystalline polymer. Low frequency Raman scattering revealed little change in the thickness of the lamella during degradation.
The continuous barrier of the crystalline lamellae, and its barrier to oxygen diffusion into the plastic, is replaced with fragmented nano-domains which should afford a less effective barrier to oxygen permeation into the plastic.
This change further catalyses the further degradation of the material by oxidation.