November 15, 2019 report
Using aluminum and lasers to make bendable glass

An international team of researchers has found a way to make bendable glass using lasers fired at crystalline aluminum oxide. In their paper published in the journal Science, the group describes their technique and the features of the glass they produced. Lothar Wondraczek with the University of Jena has published a companion piece in the same journal issue outlining the history of scientists attempting to overcome the brittleness of glass.
Glass is somewhat strong, but only up to a point; it is also very brittle. If you drop a drinking glass, it will likely shatter on the floor. As Wondraczek notes, scientists have been searching for ways to make glass less brittle for as long as people have been making glass. Bendable glass would mean drinking glasses that survive a fall, or smartphone screens that do not crack. In this new effort, the researchers say they have taken a step toward that goal.
Ordinary glass is made from silica and oxygen, and it is known as an amorphous solid—a state in which a material's molecules are locked together—in the case of glass, in a random fashion. It is transparent because photons can pass through it without interacting with any of the electrons in the glass. In this new effort, the researchers used crystalline aluminum oxide instead of sand to make some tiny glass samples. To do so, they fired intense bursts of laser light at a sample to turn it into a purple plasma. The material was then allowed to cool on a substrate.
Testing of the resulting material (sheets 60 nanometers thick and two micrometers wide) showed it to be transparent and far less brittle than ordinary glass. The sheets were also bendable and stretchable. The researchers found they could stretch them up to 8 percent and compress them to half their length.
The researchers also took a close look at their bendable glass using an electron microscope. Using what they found, they created computer simulations of the material they had created to better understand its properties. The model showed that the glass had a very tightly packed network of atoms that was defect free, making it bendable. Its atoms were able to switch places when exposed to pressure.
-
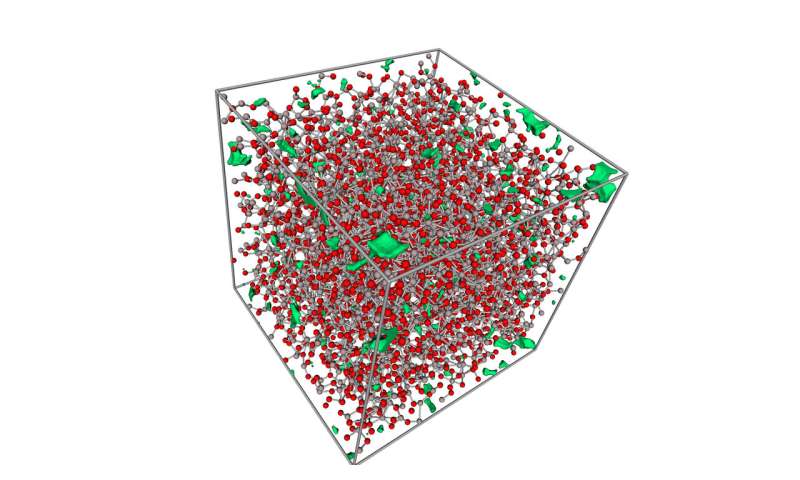
The supercomputer simulations on the atomic structure of amorphous aluminum oxide show that the atom network has very few flaws (highlighted in green), which allows the ductility mechanisms to activate without fracture. Red atoms are oxygen, grey atoms are aluminum. Credit: Janne Kalikka -

Transmission electron microscope and samples holder used to study the glass plasticity. Credit: Lucile Joly-Pottuz
More work is required before the bendable glass can be commercialized—it is still not clear if the process could be used to make bigger sheets of glass, or if it is even amenable to manufacturing.
Journal information: Science
© 2019 Science X Network