Scientists show the vulnerability of a promising two-dimensional semiconductor to air, and discover new catalyst

For the first time ever, an international team of scientists from NUST MISIS, the Hungarian Academy of Sciences, the University of Namur (Belgium), and Korea Research Institute for Standards & Science has detailed the structural changes of two-dimensional molybdenum disulfide under long-term environmental impact. The new data narrow the scope of its potential application in microelectronics and at the same time opens up new prospects for the use of two-dimensional materials as catalysts. The research results have been published in the international scientific journal Nature Chemistry.
Molybdenum disulfide (MoS2) is considered promising for a variety of microelectronic devices such as high-frequency detectors, rectifiers and transistors, so research teams around the world are actively studying its two-dimensional format, MoS2 nanofilm. However, the new study demonstrates that when this two-dimensional material is significantly oxidized in air, it turns into another connection.
Any electronic device using MoS2 without proper protection would stop working relatively quickly. To use MoS2 in microelectronics, the devices would have to be encapsulated.
"For the first time ever, we have managed to experimentally prove that a single-layer molybdenum disulfide strongly degrades under environmental conditions, oxidizing and turning into a solid solution MoS2-xOx,. The functions of a two-dimensional semiconductor without defects and losses can be implemented with molybdenum diselenides, another material with a similar structure," said Pavel Sorokin, head of the research team and leading researcher at the NUST MISIS Laboratory of Inorganic Nanomaterials.
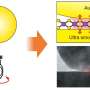
In the experiments, two-dimensional layers of molybdenum disulfide, obtained from the stratification of molybdenum disulfide crystals by ultrasound, were maintained in environmental conditions at normal room temperature and lighting for long periods (more than 18 months), during which scientists observed the changes in the structure of its surface.
"Thanks to the use of tunneling microscopy, we were able to track the structural changes of crystals of two-dimensional sulfur disulfide at the atomic level during long-term exposure to environmental conditions. We have discovered that the material previously considered stable is actually subject to spontaneous oxidation, but at the same time, the original crystal structure of MoS2 monolayers retains formations of MoS2-xOx solid solutions. Our simulations have allowed us to propose a mechanism of forming such solid solutions, and the results of the theoretical calculations are in complete agreement with our experimental measurements," said Zakhar Popov, one of the co-authors of the study and a senior researcher at the NUST MISIS Laboratory of Inorganic Nanomaterials.
"The study's second key discovery is the new material that the monolayer of the molybdenum disulfide turns into is a two-dimensional crystal of a solid solution MoS2-xOx, which is an effective catalyst for electromechanical processes," concluded Sorokin.
More information: János Pető et al, Spontaneous doping of the basal plane of MoS2 single layers through oxygen substitution under ambient conditions, Nature Chemistry (2018). DOI: 10.1038/s41557-018-0136-2
Journal information: Nature Chemistry
Provided by National University of Science and Technology MISIS