Research could minimize unwanted side effects in new drugs
Opioids relieve pain, but can cause respiratory failure and death through overdosing. Antipsychotic drugs can help people cope with mental illnesses, but muscle spasms can be a debilitating side effect. What if we could design drugs that retain their desirable traits with fewer troubling downsides?
Ron Dror, an associate professor of computer science at Stanford, led an international research team whose study – published May 2 in Nature – combines computer simulations with laboratory experiments to explore just that. The team focused on the molecular pathways within cells that control both the good and bad effects of roughly half of all common medications.
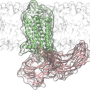
The proteins in question, known as G-protein-coupled receptors or GPCRs, sit on the exterior of cells, awaiting biochemical signals that tell cells how to behave. Those signals are normally biological molecules circulating in the blood, but can also include many drugs. When one of these signals latches onto a GPCR, it kicks off a series of molecular changes within the cell that control a wide range of functions – turning genes on or off, say, or blocking the activity of other proteins.
The Nature paper focuses specifically on the relationship between GPCRs and a family of molecules called arrestins, which are among the proteins GPCRs activate. That interaction triggers the beneficial effects of many drugs, but it also results in detrimental side effects for others.
"We want the good without the bad – more effective drugs with fewer dangerous side effects," Dror said. "For GPCRs, that often boils down to whether or not the drug causes the GPCR to stimulate arrestin."
Molecular dances
Researchers have long known that GPCRs have two parts – a fuller, rounder core and a long, narrow tail. The thought had been that the tail is responsible for activating arrestin.
Dror's team has upended that assumption by developing computer models sophisticated enough to simulate many different possible interactions between GPCRs and arrestins. These models showed that arrestins can be stimulated by either the core or the tail.
"This is true even though the GPCR core and tail bind at completely different interfaces on the arrestin," Dror said, "and the core and tail together can activate arrestin even more."
The team confirmed several computational findings through experiments performed in the lab of Martha Sommer, a molecular biologist with Charité-Universitätsmedizin Berlin. Sommer had created arrestin molecules that light up when they change shape. With these light signals, her team measured the extent to which GPCR's core, tail, or both together stimulated arrestin.
Action molecules
Dror said computer simulations like the ones he did can help guide and accelerate biological experiments. Traditional ways of imaging molecular structures capture the equivalent of still photographs. But biomolecules like GPCR and arrestins are action molecules. They bend and fold when stimulated, and it is by virtue of their dynamic interactions that they produce their various effects. A still image doesn't tell the whole story.
"With simulation, we can take GPCRs and arrestins and pull or push certain parts to see what happens. You can actually see how the atoms are moving," Dror said.
The team thinks the core might select which molecule to stimulate – arrestins or one of the many other molecules it can activate – by adopting different shapes. Taking the idea one step further, the findings suggest that drugs designed to bind to a GPCR could aim it at desired effects or block unwanted effects by influencing the shape of its core.
Related work with Dror's collaborator Mark von Zastrow at the University of California, San Francisco, also published May 2 in Nature, found that GPCRs modify the shape of arrestins so they continue signaling independent of the receptor – a never-before-seen type of arrestin signaling that von Zastrow has dubbed "kiss-and-run." Together, the findings suggest that scientists might be able to fine tune drugs to be more selective in how they activate the arrestin pathway to get the desired effects.
Dror emphasized that these studies address only a small piece of the grand puzzle whose solution could enable the design of better medicines for countless diseases. He hopes that complementary computer simulations and laboratory experiments can help address the remaining pieces.
"These behaviors are critical to drug effects, and this should help us in the next phases of our research as we try to learn more about the interplay of GPCRs and arrestins and, potentially, new drugs."
More information: Naomi R. Latorraca et al. Molecular mechanism of GPCR-mediated arrestin activation, Nature (2018). DOI: 10.1038/s41586-018-0077-3
Kelsie Eichel et al. Catalytic activation of β-arrestin by GPCRs, Nature (2018). DOI: 10.1038/s41586-018-0079-1
Journal information: Nature
Provided by Stanford University