Even bacteria have baggage—and understanding that is key to fighting superbugs
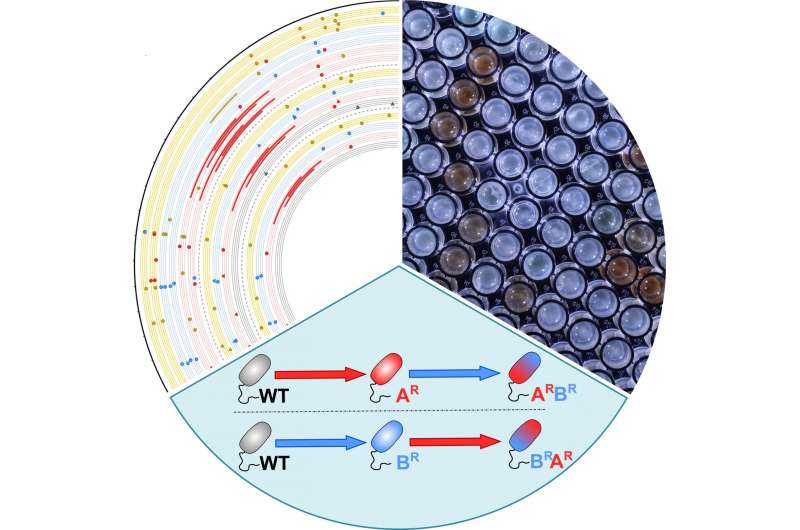
New research points to treatment strategies for multi-drug antibiotic resistance using currently available drugs. The study, publishing August 8 in the open access journal PLOS Biology by Phillip Yen and Jason Papin at the University of Virginia demonstrates how different adaptation histories of bacterial pathogens to antibiotics leads to distinct evolutionary dynamics of multi-drug resistance. In an era where there are few new antibiotics in the R&D pipeline and bacteria are developing resistance to the drugs now available, exploiting bacteria's past may be a major breakthrough for the future.
Bacteria can adapt, survive and grow in the presence of antibiotics. That's well understood, and many studies have investigated how bacteria evolve to become resistant to single drugs. The evolution of multi-drug resistance is a more complex process. New research provides understanding on how bacteria survived in the past, as they adapted to other drugs and other environments, influences how they respond and thrive under future conditions.
The study used the human pathogen Pseudomonas aeruginosa, giving the bacterial populations the chance to evolve resistance to different two-drug sequences of three antibiotics that are commonly used in clinical practice. The authors monitored the evolution of resistance over time and sequenced the bacteria's genomes to help understand the genetic basis for the resistance. Comparison of the results suggests that two different populations of bacteria may develop higher or lower levels of resistance to an antibiotic (relative to each other) if they had already adapted to different drugs in the past in a drug-specific manner.
Here the experiments were done in a test tube-like setting. Further work and clinical studies will test the clinical applicability of these history-dependent effects. Yen and Papin hypothesize that antibiotic deployment strategies that can account for how bacteria survived in the past (using clues such as patient treatment records and antibiograms) could be used to predict how they will adapt in the future. This strategy will make much better use of existing drugs by informing which drugs might lead to multi-drug resistance (and hence should be avoided), and which drugs may limit or reverse the evolution of antibiotic resistance, depending on the bacteria's history.
And in an era where there are few new antibiotics in the R&D pipeline, exploiting bacteria's past "baggage" may be a major breakthrough for the future.
More information: Yen P, Papin JA (2017) History of antibiotic adaptation influences microbial evolutionary dynamics during subsequent treatment. PLoS Biol 15(8): e2001586. journals.plos.org/plosbiology/ … journal.pbio.2001586
Journal information: PLoS Biology
Provided by Public Library of Science