January 16, 2017 report
A novel way to put flame retardant in a lithium ion battery
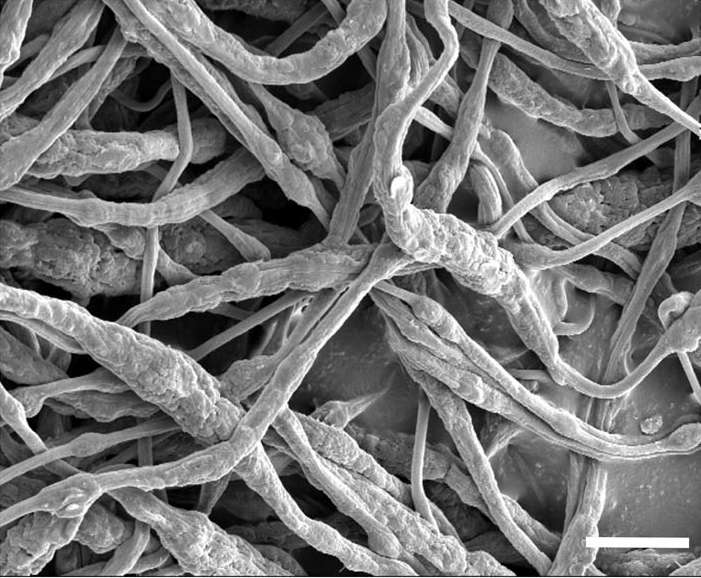
(Phys.org)—A team of researchers at Stanford University has found a novel way to introduce flame retardant into a lithium ion battery to prevent fires from occurring. In their paper published in the journal Science Advances, the team describes their technique and their results when testing it.
Reports of phones and hoverboards catching fire due to short circuits in batteries have caused alarm in the personal electronics industry—both by users and those that make the devices. Unfortunately, up until now, engineers have not been able to solve the problem completely. Most such efforts involve re-engineering devices to prevent short-circuiting and thus overheating, or attempting to put flame retardant directly in the batteries. Neither approach has proven to be entirely satisfactory. Re-engineering does not always solve the problem and the addition of flame retardant greatly reduces battery efficiency. In this new effort, the researchers describe an approach that thus far appears to offer some help—it does not stop overheating from occurring, but it is able to prevent fire.
The new approach involves encapsulating a common flame retardant called triphenyl phosphate in an extremely tiny sheath made of plastic fibers and then inserting several of them into the electrolyte that sits between the anode and cathode. The sheath keeps the retardant from actually coming into contact with the electrolyte material, which is flammable and the source of most battery fires. But the plastic fibers in the sheath have a melting point of 160° Celsius—if that temperature is reached, the plastic melts and the retardant is released into the electrolyte quashing a potential fire.
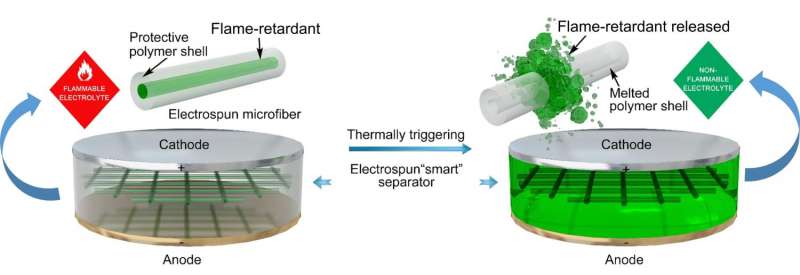
In test devices using their encapsulated flame retardant, the researchers report that the sheaths melted and the retardant was released and merged with the electrolyte in just 0.4 seconds and because of that fires were averted.
In practice, it is presumed that such an occurrence in a device would initiate a hardware error before the battery stopped working to alert a user to what had occurred. Thereafter, a user would also presumably have to purchase a new battery in order to continue using their device which would survive the overheating event.
-

GIF animation showing the EC/DEC electrolyte is highly flammable. Credit: Liu et al. Sci. Adv. 2017;3:e1601978 -
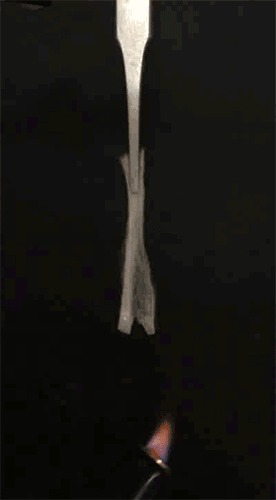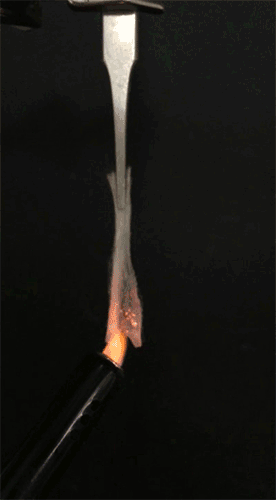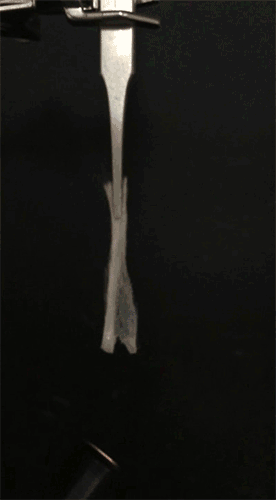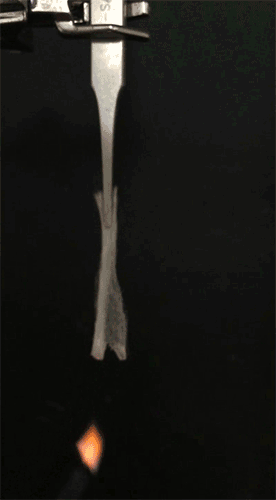
GIF animation showing the combustion of EC/DEC electrolyte with the flame-retardant TPP. Credit: Liu et al. Sci. Adv. 2017;3:e1601978 -

GIF animation showing the flammability of the EC/DEC electrolytes in the presence of the TPP@PVDF-HFP separator. The flames of the electrolyte diminish rapidly and are completely extinguished within 0.4 seconds. Credit: Liu et al. Sci. Adv. 2017;3:e1601978 -
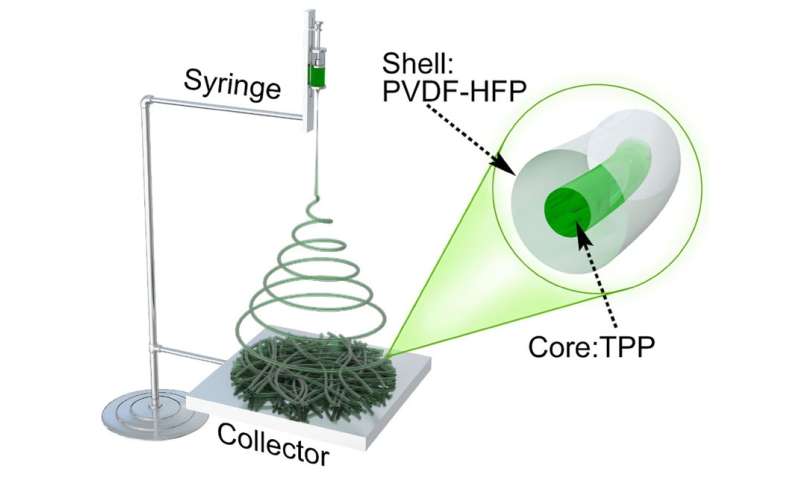
Schematic illustration for the fabrication of the microfibers by electrospinning. Credit: Liu et al. Sci. Adv. 2017;3:e1601978
More information: Kai Liu et al. Electrospun core-shell microfiber separator with thermal-triggered flame-retardant properties for lithium-ion batteries, Science Advances (2017). DOI: 10.1126/sciadv.1601978
Abstract
Although the energy densities of batteries continue to increase, safety problems (for example, fires and explosions) associated with the use of highly flammable liquid organic electrolytes remain a big issue, significantly hindering further practical applications of the next generation of high-energy batteries. We have fabricated a novel "smart" nonwoven electrospun separator with thermal-triggered flame-retardant properties for lithium-ion batteries. The encapsulation of a flame retardant inside a protective polymer shell has prevented direct dissolution of the retardant agent into the electrolyte, which would otherwise have negative effects on battery performance. During thermal runaway of the lithium-ion battery, the protective polymer shell would melt, triggered by the increased temperature, and the flame retardant would be released, thus effectively suppressing the combustion of the highly flammable electrolytes.
Journal information: Science Advances
© 2017 Phys.org



















