Physicist propose important medical applications of the magnetocaloric effect

A group of Russian physicists and Swiss colleagues have developed a way to use magnetocaloric heating or cooling of tissues for possible therapeutic interventions. They've published their results in the latest issue of the International Journal of Refrigeration.
A team of the Lomonosov Moscow State University scientists proposed a new way to use the magnetocaloric effect for the targeted delivery of drugs. Physicist Vladimir Zverev claims that this is a unique method that uses a negative magnetocaloric effect.
The gist of the magnetocaloric effect (MCE) is that when exposed to an external magnetic field, magnetic material changes temperature, rising or falling depending on the material. This phenomenon was discovered in the 19th century, though it was described only in 1917. Over the past century, MCE has not been extensively studied, interest has increased dramatically in recent decades. This is partly due to advances in the physics of magnetic materials and a growing number of possible applications. It can be used in low-temperature physics, for the production of heat engines, refrigeration and so on.
However, the majority of these applications are not ready for commercial use, mainly due to the nascent stage of the technology. For instance, although domestic magnetic refrigerators are being pursued by many scientific and industrial laboratories around the world, according to Zverev, such refrigerators would likely be very expensive.
"For such a refrigerator, a magnetic field of around one Tesla is required, which using today's technology, makes the prices very high and therefore commercially unacceptable—the very device to generate such a field will cost at least $1500," Vladimir Zverev says, adding that prices should fall as the technology advances.
However, this did not prevent the authors from suggesting a new medical application of the magnetocaloric effect, almost ready for widespread use.
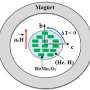
One of the methods is called "magnetic fluid hypothermia," and consists of heating cancer tumors with special magnetic nanoparticles delivered directly to the tumor site. To do this, the researchers developed a unique tool to create an alternating, high-frequency magnetic field. First, the primary research of various cancerous cell cultures was conducted. The researchers also conducted studies on mice, which proved biocompatibility and non-toxicity of the microparticles. Experiments on the microparticles' pharmacokinetics were conducted, demonstrating internal dynamics like deliverability to the tumor, spreading in the body with the blood flow, etc.
Another application is in the area of medical implants. Rejection is one of the problems with such artificial implants as joints, abdominal nets, stents, urinary and biliary ducts, etc.. The authors propose applying a special coating to implants consisting of several layers. The first layer is a magnetic material that is cooled in an external magnetic field (a material with a negative magnetocaloric effect). This layer may be a thin film or a suspension of magnetic microparticles. The second layer is the polymer matrix that, like a sponge, absorbs the drug. The polymer matrix is in direct thermal contact with the magnetocaloric material. This entire structure is placed in the body during the operation.
The polymer used in the technology behaves like a jelly at normal body temperature, and holds the drug inside. When the magnetic field lowers the temperature, the polymer transits in a liquid state and releases drug at the site of the implantation. If inflammation occurs after insertion of the implant, the non-invasive application of an external magnetic field (for example, in MRI) allows the release of the desired drug dosage at a targeted time and place.
This method of targeted drug delivery only affects the source of inflammation, leaving the rest of the body unaffected. However, it is unclear what to do if the coated drug is depleted.
Zverev says that this problem is solvable: "First, in some cases just a single drug input is needed—for example, to paste the abdominal mesh. Release dosage of the drug can be controlled by regulating the magnitude of the external magnetic field. It is also possible to replenish the coat, since a drug may be chemically linked to the magnetic particles which can be 'dragged' to the desired location in the body by an external magnetic field. This method is still theoretical, however."
More information: A.M. Tishin et al, A review and new perspectives for the magnetocaloric effect: New materials and local heating and cooling inside the human body, International Journal of Refrigeration (2016). DOI: 10.1016/j.ijrefrig.2016.04.020
Provided by Lomonosov Moscow State University