June 6, 2016 report
New nanoparticle-based contrast agent that is pH dependent
(Phys.org)—A new type of nanoparticle-based MRI contrast agent demonstrates selectivity for tumor cells over non-cancerous cells, can detect hypoxia, and is sensitive enough allow for the detection of difficult-to-find cancer cells according to a study in Nature Nanotechnology.
Researchers from the University of Tokyo, Tokyo Institute of Technology, Kawasaki Institute of Industry Promotion, and the Japan Agency for Quantum and Radiological Science and Technology have developed a contrast agent from calcium phosphate-based nanoparticles that release a manganese ion an acidic environment. The manganese ion then binds to proteins allowing for a stronger contrast signal that lasts longer than current clinically approved alternatives. These nanoparticle-based contrast agents are also functionalized with a poly(ethylene glycol) shell that promotes attachment to the cancer cell.
Magnetic resonance imaging relies on the excitation and subsequent relaxation of protons. In clinical MRI studies, the signal is determined by the relaxation time of the hydrogen protons in water. To get a stronger signal, scientists can use contrast agents to shorten the relaxation time of the protons.
MRI is non-invasive and does not involve radiation, making it a safe diagnostic tool. However, its weak signal makes tumor detection difficult. The ideal contrast agent would select for malignant tumors, making its location and diagnosis much more obvious.
Nanoparticle contrast agents have been of interested because nanoparticles can be functionalized and, as in this study, can contain various metals. Researchers have attempted to functionalize nanoparticles with ligands that attach to chemical factors on the surface of cancer cells. However, cancer cells tend to be compositionally heterogeneous, leading some researchers to look for nanoparticles that respond to differences in pH or redox potential compared to normal cells.
Peng Mi, Daisuke Kokuryo, Horacio Cabral, Hailiang Wu, Yasuko Terada, Tsuneo Saga, Ichio Aoki, Nobuhiro Nishiyama, and Kazunori Kataoka developed a contrast agent that is comprised of Mn2+- doped CaP nanoparticles with a PEG shell. They reasoned that using CaP nanoparticles, which are known to be pH sensitive, would allow the targeted release of Mn2+ ions in the tumor microenvironment. The tumor microenvironment tends to have a lower pH than the normal regions to rapid cell metabolism in an oxygen-depleted environment. Manganese ions were tested because they are paramagnetic, which makes for a good contrast agent. They also bind to proteins creating a slowly rotating manganese-protein system that results in sharp contrast enhancement.
Studies with CaPMnPEG nanoparticles showed that the nanoparticles are stable under physiological conditions (pH 7.4) and only 8% of the Mn2+ ions were released under these conditions. In conditions that mimic the tumor microenvironment and intercellular environment, within four hours 36% of the Mn2+ ions were released at pH 6.7, 71% at pH 6.5, and over 90% at pH 5.
Additionally, tests with CaPMnPEG and human serum albumin (HSA) in an acidic medium showed significant signal amplification. This is due to protein binding of the Mn2+ ion, which greatly increased the ion's molecular relaxivity.
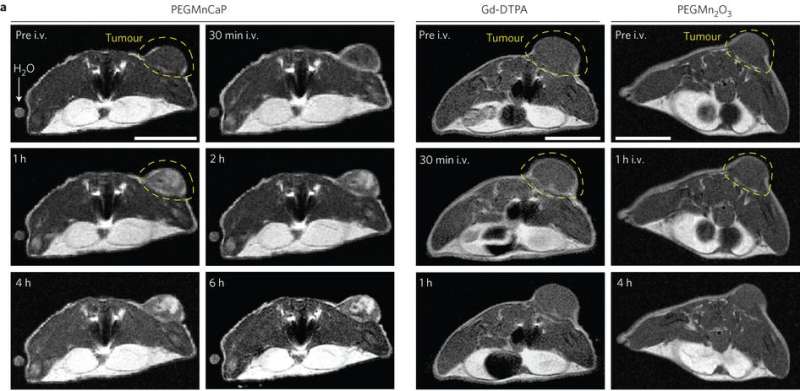
These results were promising, so Peng Mi, et al. then tested whether the CaPMnPEG contrast agent worked in solid tumors. Because Mn2+ remains confined within the nanoparticle matrix at physiological pH, CaPMnPEG demonstrate a much lower toxicity compared to MnCl2. MRI studies showed a tumor-to-normal contrast of 131% after 30 minute, which is much higher than Gd-DTPA, a clinically approved contrast agent. After an hour, the tumor-to-normal ratio was 160% and remained around 170% for several hours.
Three-dimensional MRI studies of solid tumors showed that without the addition of CaPMnPEG, only blood vessels were visible. However, upon adding CaPMnPEG, the tumor was easily distinguishable. Additionally, there is evidence that excess Mn2+ leaves the plasma after an hour. The contrast signal remained strong for several hours indicating that protein binding rather than Mn2+ concentration is important for signal enhancement.
Notably, there were brighter contrast enhancement regions on the tumor. Additional experiments demonstrated that higher contrast regions correlated to tumor hypoxia. Malignant tumors tend to be hypoxic leading to a buildup of lactate. This results in lowering the pH. Other techniques to detect hypoxia tend to only look at the surface of the target tissue or, in the case of PET, involve radiation and poor resolution. This technique is safer, non-invasive, and not confined to the surface tumors.
Finally, tests with metastatic tumors in the liver (C26 colon cancer cells) showed that CaPMnPEG works well in solid organ analysis and is highly sensitive to detecting millimeter-sized micrometastasis. Unlike other contrast agents used in the clinic, CaPMnPEG provided a contrast signal that lasted for several hours after injection. After an hour, the signal was enhanced by 25% and after two hours, the signal was enhanced by 39%.
This research reports the development and characterization of a promising new contrast agent that may provide better tumor detection and diagnosis. Additional studies could involve testing this contrast agent with other diseases that cause physiological changes in pH as well as in vivo monitoring of new drugs.
More information: Peng Mi et al. A pH-activatable nanoparticle with signal-amplification capabilities for non-invasive imaging of tumour malignancy, Nature Nanotechnology (2016). DOI: 10.1038/nnano.2016.72
Abstract
Engineered nanoparticles that respond to pathophysiological parameters, such as pH or redox potential, have been developed as contrast agents for the magnetic resonance imaging (MRI) of tumours. However, beyond anatomic assessment, contrast agents that can sense these pathological parameters and rapidly amplify their magnetic resonance signals are desirable because they could potentially be used to monitor the biological processes of tumours and improve cancer diagnosis. Here, we report an MRI contrast agent that rapidly amplifies magnetic resonance signals in response to pH. We confined Mn2+ within pH-sensitive calcium phosphate (CaP) nanoparticles comprising a poly(ethylene glycol) shell. At a low pH, such as in solid tumours, the CaP disintegrates and releases Mn2+ ions. Binding to proteins increases the relaxivity of Mn2+ and enhances the contrast. We show that these nanoparticles could rapidly and selectively brighten solid tumours, identify hypoxic regions within the tumour mass and detect invisible millimetre-sized metastatic tumours in the liver.
Journal information: Nature Nanotechnology
© 2016 Phys.org