Discovery suggests possible revolutionary antibiotics
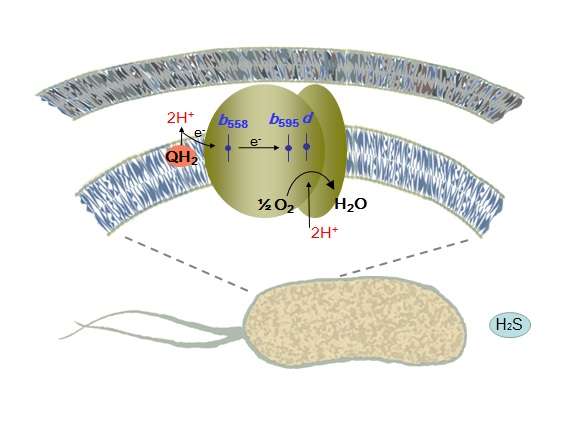
An international team of including the Lomonosov Moscow State University researchers have determined which enzyme enables Escherichia coli bacterium (E. coli) to breathe. The study is published in the Scientific Reports.
Scientists discovered how the E. coli bacterium can survive in the human gut, resolving the mystery of how they breathe. Vitaliy Borisov, senior researcher, explains that E. coli uses special enzymes that are absent in the human body. This means that the discovery can contribute to the creation of new drugs, which would be detrimental to the bacteria without harming a human.
The energy for the vital activity of any organism comes from food, and is generated by the means of redox processes in the body. The food is converted into energy—not directly, but through intermediaries. First, the complex molecules are decomposed into simpler ones: Proteins are decomposed into amino acids, fats to fatty acids, carbohydrates to monosaccharides. Oxidation of simpler molecules releases energy, which all is contained in the electrons.
The electrons pass to the respiratory chain with the so-called reducing equivalents (electron-carrying compounds). They are NADH (nicotinamide adenine dinucleotide) and ubiquinol, also known as coenzyme Q. These two basic reducing equivalents are engaged with the processing of food— NADH is a water-soluble compound and ubiquinol is fat-soluble. Membranous enzymes accept electrons from reducing equivalents and transfer them to molecular oxygen.
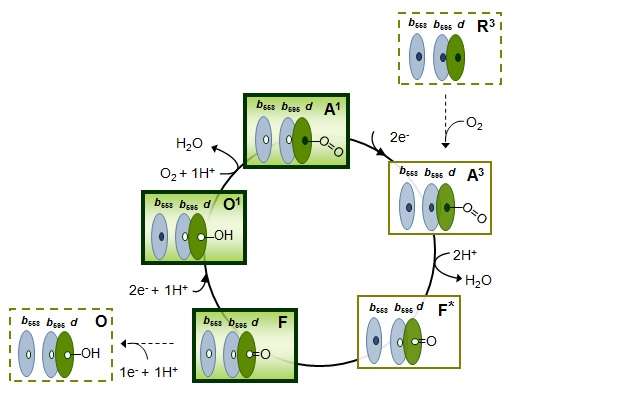
The terminal cytochrome oxidase is the main membrane enzyme responsible for human mitochondrial respiration and was thought to be used for the breathing of E. coli as well. The oxidase action functions simply, transferring electrons to molecular oxygen, after which reducing equivalents are oxidized again, and as a result, "the energy currency" of the cell, the proton-moving force, is generated.
'If you stop breathing, you die just because oxygen does not flow to the oxidase, and it does not produce energy,' says Vitaly Borisov.
The Escherichia coli bacterium lives in the gastrointestinal tract, where a lot of hydrogen sulfide is produced, which attenuates mitochondrial respiration. Free hydrogen sulfide inhibits cytochrome oxidase work. Its concentration exceeds several hundred times the minimum concentration required for substantial blocking of this enzyme. Hence, it seems that the E. coli bacterium cannot "breathe," but despite that, the bacteria survive in the intestine. The researchers assumed that the respiration in the presence of hydrogen sulfide is still possible via another oxidase. But obviously, human and bacterial respiration processes are different. Each cell in our body "breathes" due to the work of only the cytochrome-c oxidase. However, the E. coli bacteria has two types of oxidase: bo-type cytochrome oxidase (an analogue of "human" cytochrome-c oxidase) and completely different bd-type cytochromes.
'Our hypothesis was that the bd-type oxidase (bd-I and bd-II) are more resistant to the hydrogen sulfide inhibition than the bo-type cytochrome oxidase,' said Vitaly Borisov.
To test this hypothesis, scientists needed to learn how the sulfide presence in the environment affects the growth of the E. coli bacteria cells, which have only one terminal oxidase (bd-I, bd-II or bo) in the respiratory chain. A variety of biochemical, biophysical and microbiological methods and approaches were applied, as well as the method of the intended mutagenesis. The hypothesis was fully confirmed.
'Bo-oxidase's activity is completely inhibited by the hydrogen sulfide, while the work of the bd-oxidases remains untouched by the H2S. Thus, in order to successfully produce the main types of "energy currency" under a high concentration of hydrogen sulfide, the intestinal microflora inhabitants should use a unique type of terminal oxidase, which is missing in the cells of humans and animals,' says Vitaly Borisov.
The discovery could be used in the future to develop medicines that regulate the intestinal microflora to relieve it from harmful bacteria. As human cells do not contain the bd-type oxidase, the question of the ability to combat disease-causing bacteria without causing harm to the human body becomes relevant. For example, the bacterium causing tuberculosis, whose primarily membrane enzyme is also a bd-type oxidase, quickly gains resistance to classical antibiotics. Through this study, there is a prospect of a new type of antibiotic inhibiting oxygen supply only to the harmful bacteria cells, without affecting human cells.
More information: Elena Forte et al, The Terminal Oxidase Cytochrome bd Promotes Sulfide-resistant Bacterial Respiration and Growth, Scientific Reports (2016). DOI: 10.1038/srep23788
Journal information: Scientific Reports
Provided by Lomonosov Moscow State University




















