A surprising makeover turns an ordinary protein into a magnetic sculptor
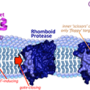
By studying an unusual group of magnetic microorganisms, scientists at UC Berkeley have uncovered a new and unexpected function for a ubiquitous protein family. Proteases are workhorse enzymes found in all living organisms that act in general cellular maintenance and communication by chewing up proteins. In a paper publishing in the Open Access journal PLOS Biology on March 16th 2016, the Komeili lab, along with collaborators in the Hurley and Chang groups, have now shown that a bacterial protein called MamO has been transformed from a common protease to an inactive enzyme that helps to build magnetic nanoparticles using a novel metal-binding motif.
Many organisms, ranging from mammals to small single-celled algae, add functionality to their cells through the construction of elaborate three-dimensional minerals. The products of these "biomineralization" processes are of great interest in both basic and industrial settings. "We would like to know how minerals are built in nature since they constitute a fundamental survival strategy for many organisms," said Dr. Komeili. In addition, scientists are interested in mimicking natural biomineralization systems to design customized nanoparticles for use in a number of applications. In order to study the biological control of mineral production, Komeili and his team have been studying how a group of microorganisms, called magnetotactic bacteria, makes chains of magnetic crystals that allow the cells to swim along the earth's geomagnetic field. Their study focuses on Magnetospirillum magneticum AMB-1, a bacterium that builds small compartments called magnetosomes, which house the machinery for crystallizing iron atoms to make magnetite. Komeili's group knew that two proteins, MamE and MamO, are required at the earliest stages of mineral formation in AMB-1. Based on predicted similarities to known enzymes in the DNA sequences for each gene, both proteins had been designated as proteases.
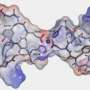
In an effort to understand details about how the protein works, David Hershey, a graduate student in the Komeili lab, wanted to understand the precise architecture and activity of MamO. They used X-ray crystallography to define the atomic structure of MamO. At first glance, MamO adopts a shape that is quite similar to that of other proteases. But by examining the structure more closely, Hershey and his colleagues found that MamO is riddled with changes that show it has lost the ability to perform its protease function. Instead, they discovered that MamO has an unexpected metal-binding activity that is required for AMB-1 to make magnetic crystals. Their results show that this ancient protease scaffold has been transformed into a novel metal-binding feature. Surprisingly, they found that a process similar to the one discovered in AMB-1 has occurred in all major groups of magnetotactic bacteria. Using the motifs they identified in MamO, they show that the genomes of these very diverse species also have inactive proteases. By tracing their evolutionary trajectory they found that the inactive proteases have arisen numerous times throughout the evolution of magnetosomes by convergent evolution. "We really thought that something this unusual would have evolved only once. That just isn't the case. It really just cements how unusual this process is," says David Hershey. Komeili and his team think that dramatic changes to the environment in the distant past provided the selective pressure that necessitated the presence of inactive proteases for formation of magnetic nanoparticles.
The unexpected findings on the structure, activity and evolution of MamO has set the stage for a whole host of future explorations of biomineralization. Komeili's group wants to continue to investigate the precise role of metal-binding by MamO in biomineralization. Does MamO directly sequester iron to build the nucleus of the magnetic crystals? Or, does it act as a monitoring system for the local magnetosome environment, initiating biomineralization at the appropriate time? More broadly, Dr. Komeili hopes that the metal-binding activity of MamO can be exploited to produce magnetic particles synthetically using a simplified chemical system.
More information: Hershey DM, Ren X, Melnyk RA, Browne PJ, Ozyamak E, Jones SR, et al. (2016) MamO Is a Repurposed Serine Protease that Promotes Magnetite Biomineralization through Direct Transition Metal Binding in Magnetotactic Bacteria. PLoS Biol 14(3): e1002402. DOI: 10.1371/journal.pbio.1002402
Journal information: PLoS Biology
Provided by Public Library of Science