Electrically induced arrangement improves bacteria detectors

Viruses that attack bacteria—bacteriophages—can be fussy:They only inject their genetic material into the bacteria that suit them. The fussiness of bacteriophages can be exploited in order to detect specific species of bacteria. Scientists from Warsaw have just demonstrated that bacteriophage-based biosensors will be much more efficient if, prior to the deposition on the surface of the bacteriophage sensor, their orientation is ordered in an electric field.
Bacteriophage-based biosensors are a promising method for detecting particular species of bacteria. The sensitivity of current sensors coated with bacteriophages—that is, viruses that prey on bacteria—is far from ideal. In the journal Sensors and Actuators B: Chemical, researchers from the Institute of Physical Chemistry of the Polish Academy of Sciences (IPC PAS) in Warsaw, Poland, have presented a method for creating layers of bacteriophages which significantly increases the efficiency of detection. This achievement paves the way for the production of low-cost biosensors capable of rapidly and reliably detecting specific species of bacteria.
The late detection and identification of bacteria is the causes of many tragedies. The lack of reliable and rapid medical tests means that doctors only find out after several hours which bacterial species is wreaking havoc in the body of the patient. As a result, instead of administering the optimal antibiotic at an early stage of the disease, they have to guess—and they often get it wrong, with disastrous consequences for the patient.
"Hospital-acquired infections, to which 100 thousand patients in the United States alone succumb each year, are just one of the problems arising from the lack of good methods for the detection of undesirable bacteria. Industrial contamination is no less important. For example, nobody wants to sell—much less buy—carrot juice with dangerous typhoid or sepsis bacteria. However, such cases continue to occur," says Dr. Jan Paczesny (IPC PAS).
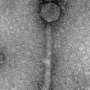
Attempts have been underway for some time to construct bacteria sensors that exploit bacteriophages. A single phage, with a length of about 200 nanometers, consists of a head (capsid) containing DNA or RNA and a tail through which genetic material is injected into the interior of the bacteria. The mouth of the tail is surrounded by fibrils. They perform a very important function as receptors detecting the presence of bacteria and recognizing their species. The bacteriophage cannot take any risks—its genetic material must reach the interior of only those bacteria that have suitably matching genetic machinery. If the phage were to inject its genetic code into the wrong bacteria, it would self-destruct rather than leveraging the bacteria for its reproduction.

The specific structure of bacteriophages means that when they are deposited on the surface, they are arranged at random, and most of them cannot effectively penetrate the space around them with their receptors in search of bacteria. As a result, only a few bacteriophages in the detection layer of current biosensors can fulfill their role and the equipment's sensitivity is greatly reduced.
"Phage heads are electrically negatively charged, whereas the filaments penetrating the surroundings are positive. The bacteriophage is therefore an electrically polarized entity. This gave us the idea of 'ordering' the bacteriophages using an electric field," says Ph.D. student Kinga Matułaa (IPC PAS).
The idea was simple, but its implementation proved to be far from trivial.
"There is a high pressure of up to 50 atm in the phage heads. This is what enables the bacteriophage to inject its genetic material. That's fine, but it means that bacteriophages like highly saline solutions, because the pressure difference between the head and the environment is reduced. Such solutions are highly conductive, and therefore the electric field inside them is present only in a thin layer at the surface—further on, it drops to zero," explains Ph.D. student Łukasz Richter.
During their experiments, the Warsaw-based scientists, led by Prof. Robert Holyst, used an appropriately selected constant electric field. Bacteriophages were deposited on a carefully constructed glass substrate, coated first with titanium and then with gold. The titanium served as the glue binding the gold with the glass, while the gold was the main 'bait' to which the bacteriophages bound. Unfortunately, not only bacteriophages like gold—so do bacteria. To prevent the binding of random bacteria with the gold layer, the empty spaces between the deposited bacteriophages were covered with a neutral protein (casein).
T4 bacteriophages that attack Escherichia coli bacteria were used to construct the new detection layer at the IPC PAS. The phages for the studies were prepared by the team of Prof. Marcin Łoś from the Department of Biology, University of Gdansk.
"Virtually all of the bacteriophages in our detection layers stand on the substrate's surface, so they can easily spread out their receptors. The situation is somewhat similar to what is seen at a rock concert, where fans often raise their hands high above their heads in unison and wave them cheerfully in all directions. We have the impression that our phages are even happier, because we try not to place them too close to each other. After all, the neighbours' receptors should not interfere with each other," says Prof. Holyst with a smile.
Meticulous laboratory tests have established that the bacteriophage layers produced using the method developed at the IPC PAS trap up to four times more bacteria than existing layers. As a result, their sensitivity is close to that of the best biosensors that use other, more time consuming and expensive, methods for the detection of bacteria.
This method of preparing layers of ordered bacteriophages has numerous advantages. The creation of an external electric field, which is necessary to put the bacteriophages in order, is not very costly. The field acts through space and therefore direct contact of the electrodes with the solution is not required. The presence of an external electric field also means there is significantly less physico-chemical interference than when current is passed through the solution. At the same time, the method is fast and universal; it can be used not only for bacteriophages but also for other electrically polarized molecules.
More information: Łukasz Richter et al. Ordering of bacteriophages in the electric field: Application for bacteria detection, Sensors and Actuators B: Chemical (2016). DOI: 10.1016/j.snb.2015.09.042
Provided by Polish Academy of Sciences