Fungus causes emerging snake disease found in Eastern US
Researchers working for the U.S. Geological Survey have identified the fungal culprit behind an often deadly skin infection in snakes in the eastern U.S. Published this week in mBio, an online open-access journal of the American Society for Microbiology, the research shows that Ophidiomyces ophiodiicola is the definitive cause of snake fungal disease (SFD), which will help researchers pinpoint why it is emerging as a threat to snake populations and how its impacts can be mitigated.
SFD joins a list of fungal diseases causing decimation to animal populations, including white-nose syndrome in bats and chytridiomycosis in frogs and amphibians. Different fungi cause the three conditions, but their potential for destruction raises concerns.
"Unlike many bacterial and viral pathogens, fungal spores can live in the environment without a host," explains Jeffrey Lorch, a microbiologist at the USGS National Wildlife Health Center in Madison, Wisconsin. "And that means that as the host population declines, the fungus can persist in the environment, which could potentially mean it could drive hosts to extinction."
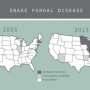
Since 2009, Lorch and his colleagues at the center have diagnosed SFD in seven species of snakes from nine different states, all in the eastern half of the U.S. In some species, such as massasauga rattlesnakes found in Illinois, the infection appears to have a mortality rate of 100%. For other species, the infection is not as deadly. "There is a fear that Ophidiomyces could drive at least some populations of snakes to extinction," says Lorch.
Although skin lesions on infected snakes often contained Ophidiomyces ophiodiicola, no one had shown the definitive laboratory proof that it was causing the disease. The USGS-led team ran those experiments by culturing O. ophiodiicola from an infected wild water snake, and then using it to inoculate five different skin sites on eight corn snakes in the laboratory. All eight snakes developed swelling and scale lesions characteristic of SFD. In contrast, none of the seven control group snakes, which were sham inoculated with saline solution, developed lesions.
The lesions appeared within 4-8 days and were more likely to form at sites that had first been rubbed with sandpaper to cause an abrasion. After about two weeks, the infected snakes had rough, brown crusty lesions and proceeded to molt. Infected snakes molted more frequently, possibly as an immune response that helps snakes shed the fungus. Two infected snakes refused food when they were experiencing severe head swelling. Infected snakes were also observed out in the open, exposed area of their enclosures twice as often as uninfected snakes.
It's not known how the skin disease causes death in wild animals, but Lorch suspects it is multifactorial. "It could be due to predation or exposure if snakes are out and about when they shouldn't be. They could be getting secondary skin infections if bacteria get in." He notes that dehydration or starvation could also pose problems for infected snakes. There is also concern that environmental factors such as climate change could be compromising the ability of wild snakes to avoid, fight off, and recover from the infections.
"We can't move forward with management of a disease in the wild if we don't know what's causing it," says Lorch. The identification of O. ophiodiicola will allow researchers and wildlife biologists to build a management plan, especially for at-risk snake populations.
Journal information: mBio
Provided by American Society for Microbiology