Portable device can quickly test for sickness-causing toxins in shellfish
Mussels, oysters, scallops and clams might be ingredients for fine cuisine, but they can also be a recipe for diarrhetic shellfish poisoning (DSP). That's a gastrointestinal illness people can get if those tasty morsels contain marine toxins. Now, researchers are reporting in ACS' Journal of Agricultural and Food Chemistry the development of a portable, inexpensive device that can quickly and easily screen freshly caught shellfish for these substances.
DSP is caused by eating shellfish that have accumulated okadaic acid (OA) or related marine toxins. Algal blooms - commonly referred to as "red tides" - can produce these substances, which shellfish can accumulate through filter feeding. Because cooking the shellfish does not destroy the toxins, several regulations are in place to prevent the sale and consumption of tainted shellfish. To comply with these regulations, the current practice is to send samples to labs that use expensive, technically intense and slow tests. Waqass Jawaid and colleagues set out to develop an inexpensive, easy-to-use and portable device that maintained the rigorous testing standards of off-site labs but could quickly test shellfish on boats and at other remote locations.
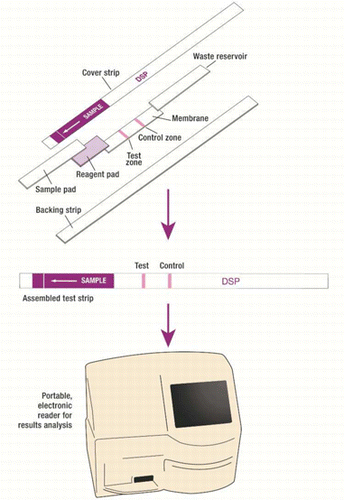
The researchers adapted a test called a lateral flow immunoassay (LFIA), which is like a home pregnancy test strip. This LFIA combines simple test procedures with an antibody previously shown to specifically bind to three OA toxins. The small, portable device can accurately screen for presence of these substances in less than 20 minutes on a boat, before it goes further into the supply chain. If the test is positive, then the shellfish would not be sold. If the LFIA readout is negative, then an additional, easy-to-use test could be conducted dockside for "total toxins," which would include detection of a fourth type of OA.
More information: Development and Validation of a Lateral Flow Immunoassay for the Rapid Screening of Okadaic Acid and All Dinophysis Toxins from Shellfish Extracts, J. Agric. Food Chem., 2015, 63 (38), pp 8574–8583, DOI: 10.1021/acs.jafc.5b01254
Abstract
A single-step lateral flow immunoassay was developed and validated to detect okadaic acid (OA) and dinophysis toxins (DTXs), which cause diarrhetic shellfish poisoning. The performance characteristics of the test were investigated, in comparison to reference methods (liquid chromatography tandem mass spectrometry and/or bioassay), using both spiked and naturally contaminated shellfish. A portable reader was used to generate a qualitative result, indicating the absence or presence of OA-group toxins, at concentrations relevant to the maximum permitted level (MPL). Sample homogenates could be screened in 20 min (including extraction and assay time) for the presence of free toxins (OA, DTX1, DTX2). DTX3 detection could be included with the addition of a hydrolysis procedure. No matrix effects were observed from the species evaluated (mussels, scallops, oysters, and clams). Results from naturally contaminated samples (n = 72) indicated no false compliant results and no false noncompliant results at <50% MPL. Thus, the development of a new low-cost but highly effective tool for monitoring a range of important phycotoxins has been demonstrated.
Journal information: Journal of Agricultural and Food Chemistry
Provided by American Chemical Society

















