Intense X-ray technology sheds new light on dynamics of how minerals, fluids interact

It's often true that to get to the bottom of big problems or understand massive systems, you have to go deep into the details.
When those details are the size of atoms and molecules, it takes special equipment and extraordinary precision to get what you're after.
A new study by a team including University of Delaware researcher Neil Sturchio and his colleagues at the Argonne National Laboratory and the University of Illinois at Chicago has given scientists an unprecedented look at such detail, showing how minerals and fluids interact in extreme conditions.
The study, to be published Friday in the journal Science, shows how the intense X-ray beam produced by a synchrotron – a particle accelerator – affects the way calcite and water interact.
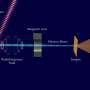
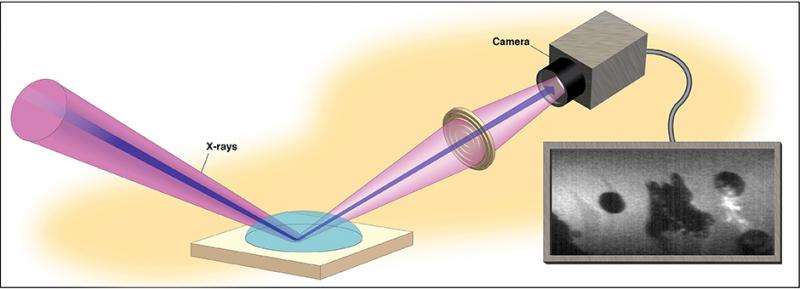
And with a new use of X-ray beams, in something called the X-ray reflection interface microscope, they also illustrated their findings with real-time topographic images of the mineral's surface.
The details they uncovered had some surprises and provided valuable information for future study.
Sturchio, who joined the UD faculty last year as professor and chair of the Department of Geological Sciences in the College of Earth, Ocean, and Environment, has studied mineral/water interactions for more than 20 years with funding from the Department of Energy.
The DOE is interested in how elements flow through groundwater and soils because those properties control how contaminants and metals are transported, with critical implications for environmental and water quality.
A 2011 tsunami-triggered nuclear accident that affected several reactors and released radioactive material in Fukushima, Japan, is a recent example of why such knowledge is essential.
Sturchio and his collaborators have demonstrated a new way of studying the microscopic structure and processes that occur where minerals and water meet, using X-ray beams both to trigger the reactions while capturing images of their effects on the mineral surface.
Usually, Sturchio said, an atomic force microscope is used for such topographic images. The new instrument – called XRIM – makes similar images at a small scale in real time while simultaneously measuring structure and composition of the mineral-water interface.
The team found that they could use the intensity of the X-ray beam as a dial to increase the dissolution rate of the calcite and control the chemistry of the solution.
The X-ray beam generates photoelectrons when it hits the crystal, Sturchio said, knocking electrons off of some of the atoms and into the solution, where they produce other reactive chemical species.
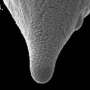
The team also observed another phenomenon when they left the X-ray beam on the mineral continuously, instead of pulsing it on and off every few seconds. The continuous beam etched the surface of the calcite, but not in the usual etch-pit shapes. Instead, it chewed out a "wormhole" in a wild shape.
"We're seeing a new phenomenon that nobody has observed before," Sturchio said, "because the only way to observe it is to watch it happening under the X-ray beam."
Scientists now can see chemical interactions where solids and liquids meet under extreme conditions.
"That is relevant to simulating conditions in a nuclear reactor," Sturchio said. "We can see what microscopic reactions will happen to the materials within the reactor, or various other materials exposed to that level of radiation."
These studies could provide critical, worst-case scenario information.
The work also gives valuable information to other researchers who now will have a better idea of how changing conditions can affect their samples.
Sturchio's work in the Department of Energy's geosciences program started in 1992 at the Argonne National Laboratory in Illinois, where he initiated this project on mineral/water interactions.
The XRIM microscope used in this study was built about three years ago, he said. An earlier version dates back about 10 years, and was invented by his Argonne colleague Paul Fenter.
The work also highlights the emerging expertise in "nanobiogeochemistry" at UD, he said, a sort of catch-all name that points to the interdisciplinary impact of the research.
"Knowing what's happening at the atomic and molecular scale makes a big difference in understanding larger-scale phenomena," he said. "You have to understand what's happening at that tiny scale to get a clearer view of the big picture."
More information: "X-ray–driven reaction front dynamics at calcite-water interfaces." Science 18 September 2015: DOI: 10.1126/science.aab3272
Journal information: Science
Provided by University of Delaware