Acetic acid as a proton shuttle in gold chemistry
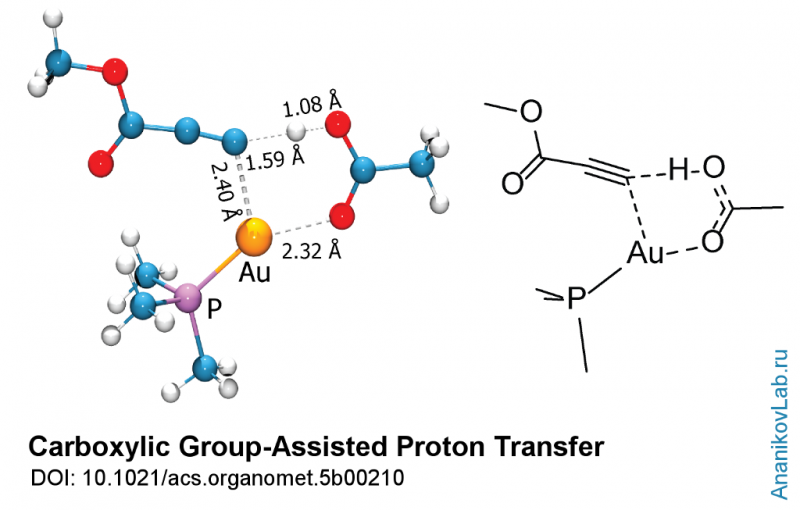
A recently published study gives a vivid example of unusual chemical reactivity associated with organogold complexes. Using modern physical methods and computational studies, the authors propose a reaction mechanism in which a molecule of acetic acid serves as a proton shuttle, transferring the hydrogen atom between the reaction centers.
Gold is one of the oldest elements known to man. The value of gold was determined by its unusual properties – heft, shine and ability to withstand oxidation and corrosion. This combination of properties determined gold use in jewelry and as a coinage metal. Ancient alchemists working with gold struggled with the chemical resistance of gold, as it does not react with concentrated acids or alkali solutions, even at high temperatures. Because of gold's chemical inertness, it is normally found in a native form and not as a part of a mineral.
Further study established that gold compounds can not only compete with traditional nickel and palladium-based catalysts in common reactions, but surpass them. Additionally, gold compounds often demonstrate novel types of reactivity compared to well-established catalysts. This allowed chemists to discover new chemical reactions and predetermined a fascinating boom in gold catalysis that we have observed in the recent years.
Professor Ananikov and co-workers introduced gold into well-known catalytic system which led to dramatic change of the reactivity and furnished the formation of novel gold-containing complexes. The complexes appeared to be air stable and were isolated in the individual state. A single crystal X-ray diffraction study ascertained the existence of a unique structural motif in the molecule, which cannot be explained within a conventional mechanistic framework.
The study was carried out using both theoretical and experimental approaches. Dedicated labeling of the reagents allowed observation of molecular reorganizations. Variation of reaction conditions helped to estimate key factors governing the transformation. In addition, computational study of the reaction provided the models of certain intermediate steps, which were invisible for experimental investigation. The theoretical data obtained was in excellent agreement with the experiment, and the researchers propose a reaction mechanism in which a molecule of acetic acid serves as a proton shuttle, transferring the hydrogen atom between the reaction centers.
The early belief in gold inactivity towards chemical transformations resulted in the fact that gold-related organometallic chemistry was developed significantly later than other coinage metals (like silver, nickel or copper). Today, the goal is to "introduce gold catalysis as a valuable practical tool in fine organic chemistry, competitive with other transition metal catalysts", says Prof. Valentine P. Ananikov.
More information: "Carboxylic Group-Assisted Proton Transfer in Gold-Mediated Thiolation of Alkynes." Organometallics, Article ASAP DOI: 10.1021/acs.organomet.5b00210
Provided by Zelinsky Institute of Organic Chemistry