May 29, 2015 report
Researchers capture higher resolution imagery of photosynthetic large light harvesting complex I
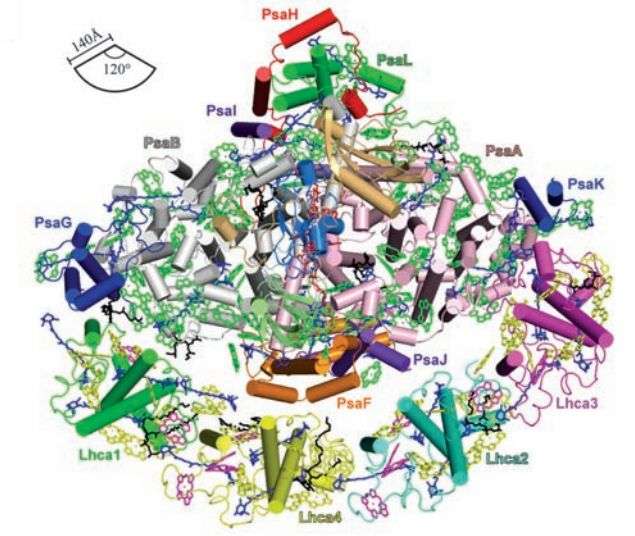
(Phys.org)—A combined team of researchers from the Chinese Academy of Sciences and Okayama University in Japan has succeeded in capturing higher resolution imagery of the large light harvesting complex I (LLHCI)—a core that surrounds photosystem I, which is one of two large pigment-protein complexes responsible for photosynthesis. In their paper published in the journal Science, Xiaochun Qin, Michihiro Suga, Tingyun Kuang and Jian-Ren Shen discuss their research and what they found when examining their shaper images. Roberta Croce of VU University Amsterdam offers a Perspectives piece on the work done by the team and what it means for better understanding how photosynthesis works.
Scientists have been studying the means by which plants convert sunlight to energy for many years, and while they have made significant progress in understanding how it works in general, they still do not really know how it works at the molecular/atomic level. To find out, they have been using tools to capture images at such levels during various parts of the process. In this new effort, the researchers built on work done by other teams attempting to capture imagery of the network of molecules that make up pigment-protein complexes.
Plants that use photosynthesis to convert sunlight to energy have two pigment-protein parts—photosystem I (PSI) and photosystem II (PSII)—the purpose of each is to capture sunlight and then to transfer the energy in it to the hub. The researchers in this effort focused on PSI, and the core that surrounds it, the LLHCI.
Prior efforts to capture imagery of LLHCI has resulted in an ability to see an overview of a network of molecules, which includes carotenoids, phylloquinones and chlorophylls, but the results have not yet been sharp enough to allow for making out the actual structure of the molecular complex. In this new effort, the researchers used X-ray crystallography on pea plants to get sharper images of LLHCI—at a resolution of 2.8 angstroms—close enough to allow them to see detailed arrangements of cofactors and pigments. Their efforts have resulted in an ability to see four additional light-harvesting complexes, providing more details on the chemical nature and geometrical arrangements of the pigments.
The work done by the researches is one more step in the long process of completely understanding photosynthesis which perhaps may one day help lead to the creation of super-efficient solar cells.
More information: Structural basis for energy transfer pathways in the plant PSI-LHCI supercomplex, Science 29 May 2015: Vol. 348 no. 6238 pp. 989-995. DOI: 10.1126/science.aab0214
ABSTRACT
Photosynthesis converts solar energy to chemical energy by means of two large pigment-protein complexes: photosystem I (PSI) and photosystem II (PSII). In higher plants, the PSI core is surrounded by a large light-harvesting complex I (LHCI) that captures sunlight and transfers the excitation energy to the core with extremely high efficiency. We report the structure of PSI-LHCI, a 600-kilodalton membrane protein supercomplex, from Pisum sativum (pea) at a resolution of 2.8 angstroms. The structure reveals the detailed arrangement of pigments and other cofactors—especially within LHCI—as well as numerous specific interactions between the PSI core and LHCI. These results provide a firm structural basis for our understanding on the energy transfer and photoprotection mechanisms within the PSI-LHCI supercomplex.
Journal information: Science
© 2015 Phys.org





















