Charge instability detected across all types of copper-based superconductors

Superconductors made of copper-oxide ceramics called cuprates are capable of conducting electricity without resistance at record-high temperatures—but still only at about one-third of room temperature. They also require cooling with liquid nitrogen, which is not practical for many potential applications, such as smart power grids, high-precision magnetometry, advanced power storage units and imaging systems.
A phenomenon called charge ordering appears to compete with superconductivity and reduce the temperature at which cuprates demonstrate superconducting properties.
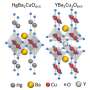
The behavior, which had been previously observed in a class of cuprates known as hole-doped cuprates, has now been detected in electron-doped cuprate superconductors for the first time. Doping involves adding impurities to the cuprate materials to produce either electrons or holes, which spur them to exhibit unusual behaviors, such as superconductivity.
The findings, published Jan. 15, 2015 online in the journal Science, suggest that charge order may be a universal feature of high-temperature superconductors. By uncovering the specific mechanism for superconductivity in cuprates, researchers hope to synthesize other materials that can superconduct at room temperature. Physicists from the University of British Columbia and the University of Maryland performed the study.
"This study's surprising results indicate that charge order must play a very important, as yet unknown, role in high-temperature superconductivity," said Richard Greene, UMD professor of physics. "The cause of high-temperature superconductivity continues to be a major unsolved question in condensed matter physics 28 years after its discovery."
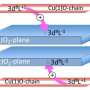
In superconductors, electrons overcome their repulsion and form pairs that move in unison and conduct electricity without resistance. In a charge-ordered state, interaction between electrons keeps them locked into a rigid pattern, which limits their ability to make the freely moving pairs required for superconductivity.
"The universality of charge ordering across very different materials shifts our perspective, and could propel future breakthroughs. We need to understand how charge ordering is formed in materials and ideally tune it, allowing superconductivity to occur at temperatures closer to room temperature," said Eduardo H. da Silva Neto, a postdoctoral fellow with UBC's Quantum Matter Institute and the Max-Planck-UBC Centre for Quantum Materials, who led the study's experimental work with former UBC Ph.D. student Riccardo Comin.
For the study, UBC researchers led by Andrea Damascelli conducted resonant X-ray scattering studies on electron-doped cuprate superconductor samples prepared by UMD physics postdoctoral fellow Yeping Jiang to confirm the presence of charge ordering.
Following the confirmation, the researchers investigated a possible prerequisite for charge ordering, and consequently the suppression of superconducting properties—the "pseudogap". This gap in the energy level of a material's electronic spectrum has been closely associated with superconductivity and has been documented to exist at temperatures just above those that give rise to superconductivity.
In the current study, the researchers found that charge ordering gradually developed in the electron-doped cuprate samples at a temperature much higher than the pseudogap, contrasting previous observations in hole-doped cuprates. The findings indicate the pseudogap is not a prerequisite for charge ordering or superconductivity in electron-doped materials. This knowledge could be an important clue to solving the 28-year-old mystery of the cause of high-temperature superconductivity, according to Greene.
"Our next experiments will be to study the doping dependence and temperature dependence of the charge order in electron-doped cuprates in the hopes of better understanding its role in high-temperature superconductivity," said Greene.
More information: "Charge ordering in the electron-doped superconductor Nd2–xCexCuO4," by E.H. da Silva Neto et al. Science, www.sciencemag.org/lookup/doi/ … 1126/science.1256441
Journal information: Science
Provided by University of Maryland