Missing electrons the secret to mine metal

Researchers have advanced the quest to understand how one of Australia and the world's largest zinc and lead mining deposits was formed.
Covering 2 km2, the Here's Your Chance (HYC) mine is located in the Middle Proterozoic McArthur Basin, in the Northern Territory.
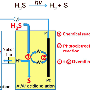
Mineral deposits like HYC's are created when sulfate in the ore fluids is reduced to produce lead and zinc sulfides, a process that requires electrons.
Yet researchers investigating the chemistry of HYC's formation say local sediments may have supplied only around one-third of the electrons required to form the deposit.
Dr Jeffrey Dick from Curtin University says his team looked at the amounts (or 'budgets') of carbon, sulfur and electrons required to form a deposit of HYC's size.
Their study accounted for possible flow and interaction between five conceptually distinct reservoirs: HYC's un-mineralised shale, the HYC ore deposit, oceanic water, hydrothermal ore-bearing fluids, and external reservoirs, included to allow for the introduction of external reduced carbon or sulfide.
Search for the missing electrons
Dr Dick's team first explored how many electrons could have been produced in-situ, by oxidation of organic matter within the mine sediments.
"Through mass balance calculations, we explored whether enough organic matter was present to allow for the necessary level of reduction," Dr Dick says.
"We found that the oxidisation of in-situ organic carbon provides at most one-third of the [electrons] needed to reduce sulfate to form the known quantity of ore minerals at HYC."
Dr Dick says this minimal contribution by in-situ organic matter suggests the introduction of another source of electrons, in the form of reduced carbon, or an alternative source of reduced sulfur.
He says the reduced sulfur could have come from deep seawater, which may have become progressively more sulfidic after 1.8 GA (1.8 billion years ago) and perhaps contacted localised sulphate in the ore fluids to form extra sulfide.
The missing electrons may also have been provided by natural gas containing significant amounts of hydrogen sulfide, a hydrocarbon reservoir in the subsurface, or hydrocarbons migrating from deep within the earth.
Researchers say evidence at HYC of aromatic hydrocarbons—thought to form deep underground at temperatures above 200oC—supports the idea of migration.
While the mystery of the missing electrons has not been definitively solved, Dr Dick says the study's investigative framework has proven valid and can help in understanding other lead-zinc deposits worldwide.
More information: "Combined sulfur, carbon and redox budget constraints on genetic models for the Here's Your Chance Pb–Zn deposit, Australia." GeoResJ, Volumes 3–4, September–December 2014, Pages 19-26, ISSN 2214-2428, dx.doi.org/10.1016/j.grj.2014.09.001.
Provided by Science Network WA