October 21, 2014 report
Testing shows billfish demonstrate bone remodeling without osteocytes
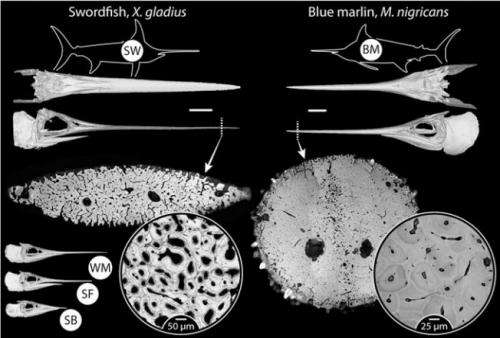
(Phys.org) —A large team of biologists with members from Israel, Germany, the U.K. and the U.S. has found that billfish are able to remodel their large protruding jawbones in the absence of osteocytes. In their paper published in Proceedings of the National Academy of Sciences, the team describes their research and results and the implications of what they found.
In humans, in order for bones to maintain their strength, they undergo a two-step process called remodeling. The first step involves tearing down damaged bone while the second involves adding new growth to replace material that is lost. Both parts of the process rely on cells known as osteocytes and together they leave behind telltale signs of re-growth known as osteons. In this new effort, the researchers sought to learn whether the bones in billfish, such as marlin and swordfish, undergo the same process to maintain bone strength.
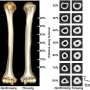
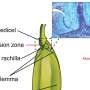
Billfish, with their long pointy bills, are very well known and prized by deep sea fishermen. But, those long bills are actually extensions of their lower jaw, which means they are bones—rostral bones, actually. Also, because the fish use the long, sword-like rostral bones for killing prey, often after high speed pursuit, the bones undergo very high stresses, which logic would suggest, means they must have some means of maintaining that strength—in its absence the bones would break and the fish would starve to death. To find out what mechanisms are at play, the researches obtained several samples from five different species of billfish, analyzed them under a microscope and then subjected them to stress tests.
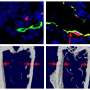
The researchers found telltale signs of remodeling, i.e. the existence of osteons in the long rostral bones but no sign of osteocytes—this suggests of course that some other unknown cells are involved in the process. They note that the osteons exhibited very high density but were an order of magnitude smaller than for similar sized mammals. The rostral bones were also particularly stiff, equal to that of bones in horses, and could take a huge amount of stress before breaking. The team's findings upend conventional thinking which has held that virtually all bone remodeling in animals involves some type of osteocytes.
More information: Remodeling in bone without osteocytes: Billfish challenge bone structure–function paradigms, PNAS, Ayelet Atkins, DOI: 10.1073/pnas.1412372111
Abstract
A remarkable property of tetrapod bone is its ability to detect and remodel areas where damage has accumulated through prolonged use. This process, believed vital to the long-term health of bone, is considered to be initiated and orchestrated by osteocytes, cells within the bone matrix. It is therefore surprising that most extant fishes (neoteleosts) lack osteocytes, suggesting their bones are not constantly repaired, although many species exhibit long lives and high activity levels, factors that should induce considerable fatigue damage with time. Here, we show evidence for active and intense remodeling occurring in the anosteocytic, elongated rostral bones of billfishes (e.g., swordfish, marlins). Despite lacking osteocytes, this tissue exhibits a striking resemblance to the mature bone of large mammals, bearing structural features (overlapping secondary osteons) indicating intensive tissue repair, particularly in areas where high loads are expected. Billfish osteons are an order of magnitude smaller in diameter than mammalian osteons, however, implying that the nature of damage in this bone may be different. Whereas billfish bone material is as stiff as mammalian bone (unlike the bone of other fishes), it is able to withstand much greater strains (relative deformations) before failing. Our data show that fish bone can exhibit far more complex structure and physiology than previously known, and is apparently capable of localized repair even without the osteocytes believed essential for this process. These findings challenge the unique and primary role of osteocytes in bone remodeling, a basic tenet of bone biology, raising the possibility of an alternative mechanism driving this process.
Journal information: Proceedings of the National Academy of Sciences
© 2014 Phys.org