February 5, 2014 report
Quantum engineering pushes refrigerator beyond classical efficiency limits
(Phys.org) —The laws of thermodynamics determine what is possible and impossible in classical systems. Lately, scientists have been working on establishing quantum analogues of these fundamental laws to determine the performance limits of quantum systems. Now in a new study, scientists have established the thermodynamic limits on quantum absorption refrigerators, and then somewhat counterintuitively show how quantum engineering techniques can push the refrigerators beyond these limits, resulting in superefficient cooling.
The findings show how quantum enhancements can allow quantum systems to exceed what is classically achievable, and marks a promising step toward the development of practical quantum cooling technologies.
The researchers, Luis A. Correa, et al., from the University of La Laguna in Spain and the University of Nottingham in the UK, have published their paper on quantum-enhanced refrigeration in a recent issue of Nature Scientific Reports.
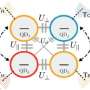
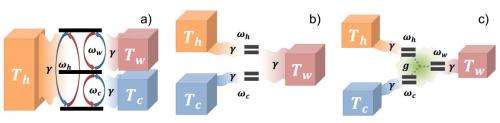
Whether classical or quantum in nature, refrigerators function by transporting energy from a cold reservoir (the object to be cooled) to a hot reservoir, usually with assistance from a power source or, in the case of an absorption refrigerator, an additional work reservoir.
"First patented by Einstein himself, absorption fridges are really 'cool,'" coauthor Gerardo Adesso at the University of Nottingham told Phys.org. "They refrigerate by absorbing heat from outside, without having to be plugged to a power socket. People use them, e.g., while camping, but these fridges have been traditionally hindered by quite a low cooling power."
For any refrigerator, the efficiency of the refrigeration process cannot exceed the Carnot limit, or else it would violate the second law of thermodynamics.
In the new study, the scientists investigated the theoretical maximum efficiency of a quantum refrigerator operating at maximum power. Efficiency at maximum power is of greater practical interest than efficiency in general, since power vanishes at high efficiencies. Here, the scientists proved that the efficiency at maximum power of a quantum refrigerator of any kind is limited by a fraction of the Carnot limit.
"Discovering that all quantum absorption fridges admit a tight model-independent performance limit was indeed surprising," Adesso said. "Establishing these bounds on efficiency at maximum power for heat engines and refrigerators has been a long-standing problem in finite-time thermodynamics."
Although this limit holds for all models of quantum absorption refrigerators, it is not the final answer. In the second part of their paper, the researchers show that quantum refrigerators can boost their performance by exploiting the system's quantum features.
Specifically, when the work reservoir is "squeezed" (an operation that allows for quantum enhancement of a system by inducing nonclassical fluctuations), the quantum refrigerator can exceed the classical performance limit and become superefficient.
"The performance bounds are just apparently violated when squeezing is applied to the heat source; since the Carnot efficiency is pushed further, the limit (relative to the Carnot) still holds," said Correa, who is at the University of La Laguna and the University of Nottingham.
The researchers explain that squeezing the work reservoir is somewhat equivalent to adding external driving, so that the work reservoir plays a more active role in the cooling process. Not only is the efficiency increased, but the total cooling power itself also increases with squeezing.
"In an absorption fridge, the hotter the work reservoir, the more powerful and efficient the refrigeration becomes," Adesso said. "Sometimes it may just not be possible to heat it up to exceedingly large temperatures even if one can afford it energetically. However, by applying quantum reservoir engineering techniques to induce squeezing, one can make the work reservoir look hotter than it actually is, thus boosting the performance of the refrigerator. This of course, has an energetic cost associated: As usual in thermodynamics, there is no free lunch."
Overall, the results have both theoretical and practical implications. For the former, they provide scientists with a glimpse of how thermodynamics looks beyond classical systems. And for the latter, reservoir squeezing could offer a practical way to improve the performance of a quantum refrigerator, with advantages over other techniques such as increasing the temperature, which may be difficult in practice. The researchers think that it will be possible to experimentally realize a superefficient quantum refrigerator with squeezed reservoir using existing technology, such as by using superconducting qubits, quantum dots, or nitrogen vacancy centers in diamonds.
"We are now thinking of experimental realizations of either absorption or minimally-controlled power-driven refrigerators, keeping an eye on possible practical applications to quantum technologies," Correa said.
"A low-consumption, high-power quantum fridge, maybe made of diamonds," Adesso added. "How cool would that be?"
More information:
Luis A. Correa, et al. "Quantum-enhanced absorption refrigerators." Nature Scientific Reports. DOI: 10.1038/srep03949
Also at arXiv:1308.4174 [quant-ph]
Journal information: Scientific Reports
© 2014 Phys.org