Ammonia: Fertiliser to be handled with care

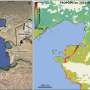
Fertiliser manufacturing has been implicated in a string of disasters prior to the deadly explosion on Wednesday that wrecked a facility in Texas.
The prime chemical in the fertiliser business is ammonia.
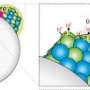
It is delivered to the soil as a colourless liquefied gas called anhydrous ("without water") ammonia, comprising 82 percent nitrogen to 18 percent hydrogen, which is absorbed on contact with the earth. Its chemical formula is NH3.
An alternative form of ammonia delivery is in granulated form as ammonium nitrate, or NH4NO3, which derives from NH3 combined with nitric acid.
Both chemicals are stable but burn at high temperatures if initiated by another source. NH3 requires additional special handling because of the risk of skin burns from exposure to the deep-chilled liquid.
NH4NO3, a soluble white powder that is readily soluble in water, becomes explosive beyond 290 degrees Celsius (554 degrees Fahrenheit)—a trigger than can come from lightning or a fire triggered by an electrical spark.
But an explosion only occurs when it is stored in large volumes, causing a chain reaction.
Ammonium nitrate is used in primitive fertiliser bombs of the kind used in the 1995 bomb attack on a US federal building in Oklahoma City, and in Oslo in 2011 by Norwegian mass murderer Anders Breivik.
Accidents involving NH4NO3 include a blast at the AZF plant in Toulouse in 2001 where 300 tonnes of the chemical were stored, killing 31 people, and in the port of Texas City in 1947 aboard a ship laden with 2,300 tonnes of it, killing nearly 600 people.
The causes of the AZF accident have never been fully clarified. Investigators suspect accidental mixing between NH4NO3 and a chloride.
In 2004, 161 people were killed and 1,300 injured in Ryongchon, North Korea, in an explosion involving train wagons carrying ammonium nitrate. Official news reports said the cause was an electrical cable that was severed during shunting.
As for NH3, the chemical becomes potentially explosive when high concentrations—between 16 to 25 percent of the atmosphere at local volumes—are ignited by a heat source greater than 1,204 F (651 C).
(c) 2013 AFP