Building blocks of life found around young star

(Phys.org)—A team of astronomers using the Atacama Large Millimeter/submillimeter Array (ALMA) has spotted sugar molecules in the gas surrounding a young Sun-like star. This is the first time sugar been found in space around such a star, and the discovery shows that the building blocks of life are in the right place, at the right time, to be included in planets forming around the star.
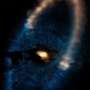
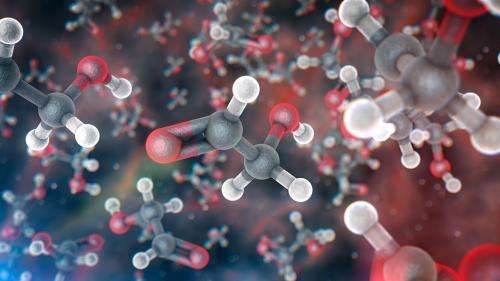
The astronomers found molecules of glycolaldehyde—a simple form of sugar—in the gas surrounding a young binary star, with similar mass to the Sun, called IRAS 16293-2422. Glycolaldehyde has been seen in interstellar space before, but this is the first time it has been found so near to a Sun-like star, at distances comparable to the distance of Uranus from the Sun in the Solar System. This discovery shows that some of the chemical compounds needed for life existed in this system at the time of planet formation.
"In the disc of gas and dust surrounding this newly formed star, we found glycolaldehyde, which is a simple form of sugar, not much different to the sugar we put in coffee," explains Jes Jørgensen (Niels Bohr Institute, Denmark), the lead author of the paper. "This molecule is one of the ingredients in the formation of RNA, which—like DNA, to which it is related—is one of the building blocks of life."
The high sensitivity of ALMA—even at the technically challenging shortest wavelengths at which it operates—was critical for these observations, which were made with a partial array of antennas during the observatory's Science Verification phase.
"What it is really exciting about our findings is that the ALMA observations reveal that the sugar molecules are falling in towards one of the stars of the system," says team member Cecile Favre (Aarhus University, Denmark). "The sugar molecules are not only in the right place to find their way onto a planet, but they are also going in the right direction."
The gas and dust clouds that collapse to form new stars are extremely cold and many gases solidify as ice on the particles of dust where they then bond together and form more complex molecules. But once a star has been formed in the middle of a rotating cloud of gas and dust, it heats the inner parts of the cloud to around room temperature, evaporating the chemically complex molecules, and forming gases that emit their characteristic radiation as radio waves that can be mapped using powerful radio telescopes such as ALMA.
IRAS 16293-2422 is located around 400 light-years away, comparatively close to Earth, which makes it an excellent target for astronomers studying the molecules and chemistry around young stars. By harnessing the power of a new generation of telescopes such as ALMA, astronomers now have the opportunity to study fine details within the gas and dust clouds that are forming planetary systems.
"A big question is: how complex can these molecules become before they are incorporated into new planets? This could tell us something about how life might arise elsewhere, and ALMA observations are going to be vital to unravel this mystery," concludes Jes Jørgensen.
More information: Research paper
Provided by ESO