Argonne scientist works to contain mercury, protect fish

(Phys.org) -- If Bhoopesh Mishra had to pick a favorite food it would be seafood. Any type of seafood. Anytime. So when as a postdoctoral scholar he had a choice to pick a research topic, his taste buds had their say.
“There was a natural connection to studying mercury contamination,” said Mishra, a guest scientist at the U.S. Department of Energy’s (DOE) Argonne National Laboratory and a faculty researcher at the Illinois Institute of Technology. “Mercury contamination is a global problem, and when it finds its way into the water, it very, very rapidly makes its way up the food chain from small fish to large fish to our dinner tables.”
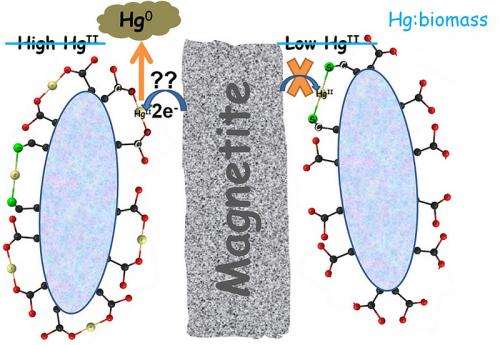
To cut back on this contamination, Mishra and fellow scientists from Argonne’s Biosciences Division identified the previously unknown process by which bacterium immobilizes toxic mercury in soil. Immobilization in soil keeps the mercury from becoming airborne and ending up in waterways where it builds up in fish. Using bacterium in this way can also reduce the natural spread of mercury and the need for large and expensive clean ups. While the research was conducted using Bacillus subtilis as a model bacterium this phenomenon is widespread among all soil bacteria.
“Airborne mercury reacts with the Earth’s ozone and hydroxyl radicals and is poured down as acid rain so you don’t have to be by any source point to have mercury contamination in rivers or lakes near you” Mishra said.
In areas of mercury contamination, such as Superfund sites, the naturally occurring ground mineral magnetite can transform mercury into a toxic, airborne form called elemental mercury. Scientists thought this conversion process was unavoidable until this study by Mishra and his co-authors. Details of how to use bacterium to prevent mercury from becoming airborne were published in the Journal of Environmental Science and Technology.
Understanding the role of bacterium in stopping the spread of mercury is the first step in creating a better system for controlling toxic mercury.
“This can have a lot of biotechnological applications,” Mishra said. “It could lead to cheaper and less invasive cleanups.”
That’s a big concern since mercury contamination exists in every region of the United States. Mercury is a major contaminant at some DOE cleanup sites and hundreds of Superfund sites.
Using the X-ray beam at the MRCAT/EnviroCAT beamline of Argonne’s Advanced Photon Source<www.aps.anl.gov/>, Mishra and his co-authors, Edward O’Loughlin, Maxim Boyanov and Kenneth Kemner, were able to pinpoint the never-before-seen mechanism by which mercury interacts with sulfhydryl groups on the bacterial cell envelope. This finding suggests that injecting organic substrates into the ground stimulates the bacteria to bind so tightly to mercury that the mercury would be unable to assume its more mobile airborne state.
To see this hidden effect of the bacteria “sequestering” the mercury through interaction with sulfhydryl groups (thiols) on bacteria, scientists had to observe low concentrations of mercury surrounded by high levels of bacteria. This observation was only made possible by the APS’s ability to pinpoint very bright X-rays on small samples.
Scientists were able to keep the mercury sequestered for two months even when a minimal amount of bacteria was available to create a protective bond. This bond held even when the mineral that can make the mercury airborne swamped it at a ratio of 500 to 1.
Mishra plans to expand on this research with follow-up studies at the APS in collaboration with scientists from Argonne and Princeton University. These studies will look at the long-term fate of sequestered mercury. There are three likely scenarios: as the original bacteria die, the mercury could revert to its base state and become airborne; another organism could metabolize the mercury, recycling it into a new toxic or non-toxic form; or the tight bond between the mercury and bacteria could keep the mercury locked in place until it slowly and naturally converts to a harmless mineral.
Based on the success of the current study, Mishra believes the mercury will stay locked in place for hundreds or thousands of years and covert to a harmless mineral. That would mean less airborne mercury to potentially land in rivers and lakes.
The U.S. Environmental Protection Agency reports that mercury contamination has been recorded in every type of fish and seafood found in natural waterways. Forty-nine states have health guidelines (water.epa.gov/scitech/swguidan … advisories/index.cfm) that recommend limits on the amount fish from natural waterways that people can safely consume per week.
Humans and animals that eat fish with mercury in it can suffer a wide variety of health problems including vision, speech and muscle impairment. Ingesting mercury-laden fish also can cause brain damage in developing fetuses. Several species of North American birds have experienced a reduction in breeding success tied to mercury poisoning from eating tainted fish.
More information: pubs.acs.org/doi/full/10.1021/es201820c
Provided by Argonne National Laboratory