Ringing the hemoglobin bell
(PhysOrg.com) -- Knowing the structure of a molecule is an important part of understanding it, but quite often it’s even more important to know how the molecule moves -- more specifically, the vibrational dynamics that drive and control its interactions with other molecules in chemical reactions. That’s particularly true of proteins, the enormously complex molecular structures found at the heart of important life processes such as cell signaling, ion transport, and other functions. But most of the available techniques for studying the vibrational properties of a protein run into some vexing limitations, especially when probing the lower frequencies at which the proteins actually do their job.
A team of researchers have found a way around that problem by using the X-ray Science Division 3-ID x-ray beamline at the U.S. Department of Energy Office of Science’s Advanced Photon Source (APS) at Argonne National Laboratory to investigate the iron-based heme molecules at the reactive core of a multitude of proteins, such as hemoglobin. Their work provides new information about the vibrational dynamics of hemes.
The experimenters from Northeastern University and Argonne employed a new technique called “nuclear resonance vibrational spectroscopy” (NRVS). “One way to use vibrational motions—particularly at high frequencies—is as a structural probe,” explains J. Timothy Sage, co-author of the article on this study that was published in the July 7, 2011, issue of Journal of Chemical Physics, where it was featured on the cover.
“People look at vibrations of individual bonds, which tells how strong the bond is.” Sage’s team had conducted previous work in the far infrared portion of the spectrum using another synchrotron x-ray facility, but found those efforts came up short: “It’s difficult in part because biological molecules always have to be put in water, and there’s a lot of competing signal from the water,” Sage said.
At the APS, the researchers could use NRVS to try another strategy. “What originally brought us to the APS was extending our investigations to lower frequencies where these vibrational motions not only are probes but actually participate in and drive the reactions that these molecules are involved in,” said Sage. The NRVS technique allows the ultimate in probe selectivity while avoiding vibrational interference from other sources. “You can zoom in right on the iron, specifically looking at one atom out of thousands of surrounding atoms, surgically targeting those vibrations.”
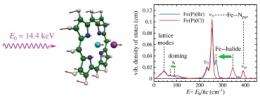
The team examined the spectrum of 57Fe vibrations in several different halide complexes of iron porphyrin molecules that mimic the heme group found in hemoglobin. In particular, they focused on a doming vibration of the heme complex that is believed to play a vital role in controlling reactions in hemoglobin such as oxygen binding.
“Just from the static structures, you can only speculate,” Sage said. “We really wanted to get something quantitative on how easy it is to excite and what the modes actually look like. These x-ray photons carry so much momentum and you can tune them so they’re targeted specifically toward the iron.” In effect, the x-ray photons “ring the bell” of the molecular structure and set it into oscillation, while allowing the vibrational spectra to be identified and studied.
Using the iron porphine molecule Fe(P) as a basis for vibrational density of states (VDOS) measurement, the experimenters used density functional theory (DFT) calculations to obtain the spectra of increasingly complex heme molecules.
“The most common biological version of this molecule [protoporphyrin IX, the actual form found in hemoglobin] is less symmetric because of these side chains sticking off the edge,” Sage said, “so the spectra look much more complicated. We spent a lot of time looking at the simpler molecule, where we could really understand precisely what the vibrations were, and then developed some mathematical analysis to find those vibrations in the more complicated molecule.”
The research team plans to both continue the present work and to expand the experimental technique to other proteins. “Most of our work has involved these heme groups, and we’ve got a really detailed understanding of how to interpret the heme molecules now,” Sage said. “So we want to extend that to look at more complicated proteins, beginning to branch out to proteins with multiple metal sites that bind iron directly to amino acid side chains without involving heme. And again, the low frequencies [available at the APS] are what is really giving us the unique window.”
Beamline 3-ID, where the experiments were conducted, has pioneered the applications of nuclear resonance techniques. Current plans include improving the energy resolution further such that collective motions of groups of atoms in the molecule can also be reliably characterized.
More information: Alexander Barabanschikov, et al. “Spectroscopic identification of reactive porphyrin motions,” J. Chem. Phys. 135, 015101 (7 July 2011). DOI:10.1063/1.3598473
Provided by Argonne National Laboratory