Turning bacteria into butanol biofuel factories
(PhysOrg.com) -- While ethanol is today's major biofuel, researchers aim to produce fuels more like gasoline. Butanol is the primary candidate, now produced primarily by Clostridium bacteria. UC Berkeley chemist Michelle Chang has transplanted the enzyme pathway from Clostridium into E. coli and gotten the bacteria to churn out 10 times more n-butanol than competing microbes, close to the level needed for industrial scale production.
University of California, Berkeley, chemists have engineered bacteria to churn out a gasoline-like biofuel at about 10 times the rate of competing microbes, a breakthrough that could soon provide an affordable and “green” transportation fuel.
The advance is reported in this week’s issue of the journal Nature Chemical Biology by Michelle C. Y. Chang, assistant professor of chemistry at UC Berkeley, graduate student Brooks B. Bond-Watts and recent UC Berkeley graduate Robert J. Bellerose.
Various species of the Clostridium bacteria naturally produce a chemical called n-butanol (normal butanol) that has been proposed as a substitute for diesel oil and gasoline. While most researchers, including a few biofuel companies, have genetically altered Clostridium to boost its ability to produce n-butanol, others have plucked enzymes from the bacteria and inserted them into other microbes, such as yeast, to turn them into n-butanol factories. Yeast and E. coli, one of the main bacteria in the human gut, are considered to be easier to grow on an industrial scale.
While these techniques have produced promising genetically altered E. coli bacteria and yeast, n-butanol production has been limited to little more than half a gram per liter, far below the amounts needed for affordable production.
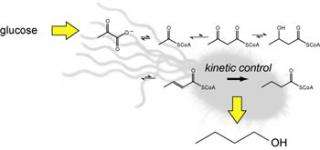
Chang and her colleagues stuck the same enzyme pathway into E. coli, but replaced two of the five enzymes with look-alikes from other organisms that avoided one of the problems other researchers have had: n-butanol being converted back into its chemical precursors by the same enzymes that produce it.
The new genetically altered E. coli produced nearly five grams of n-buranol per liter, about the same as the native Clostridium and one-third the production of the best genetically altered Clostridium, but about 10 times better than current industrial microbe systems.
“We are in a host that is easier to work with, and we have a chance to make it even better,” Chang said. “We are reaching yields where, if we could make two to three times more, we could probably start to think about designing an industrial process around it.”
“We were excited to break through the multi-gram barrier, which was challenging,” she added.

Among the reasons for engineering microbes to make fuels is to avoid the toxic byproducts of conventional fossil fuel refining, and, ultimately, to replace fossil fuels with more environmentally friendly biofuels produced from plants. If microbes can be engineered to turn nearly every carbon atom they eat into recoverable fuel, they could help the world achieve a more carbon-neutral transportation fuel that would reduce the pollution now contributing to global climate change. Chang is a member of UC Berkeley’s year-old Center for Green Chemistry.
The basic steps evolved by Clostridium to make butanol involve five enzymes that convert a common molecule, acetyl-CoA, into n-butanol. Other researchers who have engineered yeast or E. coli to produce n-butanol have taken the entire enzyme pathway and transplanted it into these microbes. However, n-butanol is not produced rapidly in these systems because the native enzymes can work in reverse to convert butanol back into its starting precursors.
Chang avoided this problem by searching for organisms that have similar enzymes, but that work so slowly in reverse that little n-butanol is lost through a backward reaction.
“Depending on the specific way an enzyme catalyzes a reaction, you can force it in the forward direction by reducing the speed at which the back reaction occurs,” she said. “If the back reaction is slow enough, then the transformation becomes effectively irreversible, allowing us to accumulate more of the final product.”
Chang found two new enzyme versions in published sequences of microbial genomes, and based on her understanding of the enzyme pathway, substituted the new versions at critical points that would not interfere with the hundreds of other chemical reactions going on in a living E. coli cell. In all, she installed genes from three separate organisms – Clostridium acetobutylicum, Treponema denticola and Ralstonia eutrophus — into the E. coli.
Chang is optimistic that by improving enzyme activity at a few other bottlenecks in the n-butanol synthesis pathway, and by optimizing the host microbe for production of n-butanol, she can boost production two to three times, enough to justify considering scaling up to an industrial process. She also is at work adapting the new synthetic pathway to work in yeast, a workhorse for industrial production of many chemicals and pharmaceuticals.
Provided by University of California - Berkeley