Unlocking the chromosome
Advances in our understanding of basic biological processes and human disease owe much to the groundbreaking research of scientists at the Chromosome Dynamics Laboratory at RIKEN Advanced Science Institute.
The DNA is a remarkable molecule in many ways. It encodes our entire genome, and if stretched out in a thin thread would measure 1.8 m in length. Yet in each and every cell, less than a tenth of a millimeter in size, the DNA must be replicated and segregated precisely in order for the cell to divide and create two daughter cells. Key to this process is the chromosome. It had widely been believed that the action of unknown protein(s) would be required in order to condense the replicated DNAs to form a chromosome. However, it wasn’t until a discovery in 1997 by Tatsuya Hirano, currently chief scientist of the Chromosome Dynamics Laboratory at the RIKEN Advanced Science Institute, that the long-sought protein machinery condensing the chromosome was finally unveiled. Hirano’s discovery of the ‘condensin’ protein complex is marked as a major breakthrough that has led to rapid advances in the elucidation of not only basic biological processes involving the chromosome but also human diseases involving chromosome aberrations.
The chromosome black box
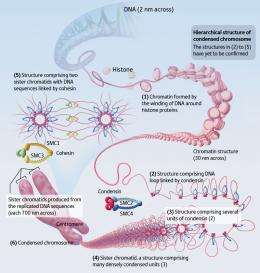
“In the early 1980s, when I entered university, I found the molecular structure of the DNA double helix on the first page of my biology textbook,” says Hirano. “The textbook also described the then recent finding that DNA wind around histone proteins to form a structure known as chromatin.” It was also known at that time that chromatin should condense further to form a chromosome (Fig. 1), a structure that had been observed by optical microscopy as early as the nineteenth century. Nonetheless, there was a huge gap in our understanding between the chromatin and the chromosomes, and the proteins involved in chromatin condensation and their mechanism of action remained a black box. “I wanted to open that black box, and this is how I became interested in the chromosome.”

Hirano entered Kyoto University’s Graduate School and joined the laboratory of Mitsuhiro Yanagida, where he engaged in chromosome research by means of yeast genetics. “My initial project was to generate lots of mutants of fission yeast. In the search for mutants displaying abnormal shapes of chromosomes, I was able to discover many genes involved in chromosome morphogenesis and segregation. During my thesis work, however, I was not completely satisfied with the genetic approach, and became interested in analyzing the chromosome using a more direct technique, namely, biochemistry. I quickly learned, though, that conventional biochemical methods do not allow us to isolate the chromosome from a cell in a very pure form.”
The discovery of condensin
In 1989, Hirano joined Tim Mitchison’s lab as a postdoctoral fellow at the University of California in San Francisco, USA, where an in vitro experimental system for chromosome assembly was under development. “Frog eggs were squashed to obtain an extract of high protein concentration. Amazingly enough, by placing the DNA in the extract, it was possible to produce the chromosome in a test tube. Because the chromosome produced in this in vitro system could be taken out in a pure form quite easily, analysis of their protein components became far easier than before,” says Hirano.
By taking advantage of this experimental system, in 1994, Hirano discovered two novel proteins essential for chromosome condensation: SMC2 and SMC4. “Just around that time, studies using budding yeast and nematodes were starting to provide evidence that some SMC-related genes are somehow involved in chromosome functions. One day my thesis advisor gave me an international call, telling me that he had also found two SMC-related genes while analyzing fission yeast mutants with morphologically abnormal chromosomes. Even more surprisingly, one of the mutants was identical to the one I discovered myself when I was a graduate student.”
Thus, the biochemical analyses of the frog chromosomal proteins by Hirano was supported by the genetic analyses of yeast and nematodes, leading him to the very general conclusion that SMC proteins play a central role in chromosome dynamics. However, this work was a mere introduction to an even larger story.
In 1995, Hirano moved to Cold Spring Harbor Laboratory on the East Coast of the USA and set up his own laboratory. In 1997, he discovered that SMC2 and SMC4 form a complex with three other proteins, and that the complex, which he named condensin, plays an essential key role in chromosome condensation. “When we examined the condensin complex under an electron microscope, we found that it had a shape like a small clip. Until that time, no one had ever seen a protein complex with such a strange shape. However, on reflection, this shape is perfectly suitable for bundling a long chromatin fiber to make a compact chromosome.”
The cohesin glue
The mid-1990s marked the dawn of genome research. In 1996, the budding yeast genome had been completely decoded and it had been revealed that there are four types of SMC in the yeast genome. “To investigate the roles of the other two types, SMC1 and SMC3, we conducted a series of experiments using a frog egg extract. We found that these two types of SMC also form a large complex with other proteins, but that the complex works in a completely different way from condensin in the process of cell division.”
From 1997 to 1998, the combined achievements of several research groups demonstrated that the complex works as a glue to link two sister chromatids. This complex is now known as cohesin (Fig. 1). “Classical research had led us to predict that there must be proteins responsible for chromosome condensation and cohesion. However, it turned out that the proteins responsible for the two processes, condensin and cohesin, contain SMC as their core components, hence sharing a structural similarity to each other. This was completely unexpected.”
Emergence and evolution of life from the viewpoint of condensin
“Even more surprisingly, condensin-like proteins were also found in prokaryotic organisms, including Escherichia coli and other bacteria, which evolved earlier than eukaryotic organisms,” continues Hirano. The replication and segregation of genetic information are fundamental to life, and while the mechanism for cell division apparently differs between prokaryotes (organisms lacking a cell nucleus) and eukaryotes (organisms bearing a cell nucleus), “the proteins responsible for condensing the chromosome were found to be common.”
Self-proliferation is regarded as one of the fundamental properties that define ‘life’. “When the primordial organism capable of replicating DNA emerged on Earth, it would have encountered a major problem of how to organize replicated DNAs and separate them into two daughter cells. The ancestor protein from which condensin originated might have appeared to resolve the problem soon after the emergence of life.”
In prokaryotes, however, no protein corresponding to cohesin has been found. “A prokaryotic condensin could serve for both condensation and cohesion, and cohesin seems to have emerged from the ancient condensin to take a differential part during evolution, perhaps around the birth of eukaryotes.”
Another noticeable finding was that higher animals and plants possess a second type of condensin. “In 2003, we discovered this new type of condensin in human cells. The original type is now called condensin I, and the second type is called condensin II. Intriguingly, condensin I and II are arranged alternatively along the axis of the chromosome. When condensin I or II is experimentally removed from the cell, the chromosome becomes morphologically abnormal in distinctive manners, indicating that the two condensin complexes have non-overlapping functions.”
Organisms with relatively small genomes, such as yeast, have condensin I only, whereas many higher animals and plants with larger genomes have both condensin I and II (Fig. 2). “Condensin II may have emerged more recently in the history of evolution. In higher animals and plants, the chromosome lengthened, so it became necessary for condensin I and II to cooperate in condensing the chromosome.”
Unlocking the black box
Hirano made the discovery of the first key to unlocking the black box of chromosome condensation. “I was lucky to have been able to discover condensin and demonstrated that it mediates chromosome condensation in 1997. But it was just a start of a long journey. Even now, the exact mechanism of chromosome condensation remains to be fully elucidated. Although a model has been proposed in which several condensin proteins linking DNA loops come together in densely arranged assemblies, we still do not know whether that model is indeed correct.”
Returning to Japan in 2006, Hirano established the Chromosome Dynamics Laboratory at the RIKEN Advanced Science Institute. “In my laboratory, we have specialists covering a broad range of research areas, including protein crystallography, biochemistry and cell biology. I want to elucidate the mechanism behind chromosome condensation by making the best use of all the techniques available. That will be my lifework.”
Of the many approaches taken by researchers at the laboratory, frog egg extract remains the most powerful experimental system. “Using this system, we can analyze the processes of chromosome condensation and segregation in great detail at an unprecedented level.”
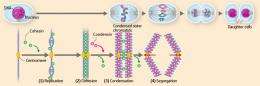
Research over the last ten years or more has firmly established that condensin and cohesin work together to support the processes of chromosome condensation and segregation (Fig. 3). Upon DNA replication, cohesin holds the replicated pair of the DNA (sister chromatids) together. When the cell prepares for division, almost all of the cohesin detaches, except for a small amount near the center of the chromosome (centromere), while condensin condenses the sister chromatids. Finally, the residual pool of cohesin detaches, and the sister chromatids are segregated and distributed to two daughter cells.
“By manipulating the level of proteins regulating the cohesin function in the frog egg extract, or adding a mutant form of the regulatory proteins with a different functionality, the action of condensin or cohesin can be altered as desired,” says Hirano (Fig. 4). “Through this type of experiments, we are endeavoring to elucidate the mechanism by which condensin and cohesin cooperate to condense and segregate the chromosomes.”
Chromosome-mediated diseases and biological phenomena
In recent years, several examples of human diseases associated with misregulation of condensin or cohesin have been reported. “Microcephaly is a disease characterized by a marked reduction in the size of the frontal lobe of the brain. A German research group examined the cells of patients with this disease and found that there is a remarkable change in the mode of chromosome condensation. We started a collaboration with that group and found that condensin II is abnormally activated in the patient cells,” says Hirano.
If condensin becomes dysfunctional and causes a condensation defect during the development of the brain, a critical gene in the formation of the brain may fail to work normally, which may in turn cause microcephaly. “Hence, it is possible that condensin is also involved in gene expression.”
The proper formation and function of an organism can only be achieved when the right genes work in the respective cells whenever necessary. Recently, the discovery that methylation of the DNA or histones affects the state of gene expression has stimulated a cascade of research in many fields of life science. “Moieties such as methyl groups control the expression of individual genes. Condensin, on the other hand, may control gene expression more globally by governing the condensation of chromosomes.”
Research into condensin and cohesin is expected to be useful in understanding the etiology of diseases involving chromosome aberrations. Normally, an individual inherits two sets of chromosomes, one from the father and the other from the mother. However, in patients with Down’s syndrome, for example, three copies of chromosome 21 are inherited, causing a range of pathological conditions. “The exact cause for abnormal inheritance of chromosomes in the patients remains unknown, but it may be that cohesin fails to work correctly on chromosome 21.”
In fact, mice deficient in cohesin have been reported to encounter an increased frequency of chromosome aberration with maternal aging. A mouse model for Down’s syndrome could therefore contribute to clarifying the etiology of this disease and the development of therapies. “Our team is also working on meiosis using the mouse as a model organism. It is known that the probability that the chromosome is inaccurately distributed in meiosis is ten times higher in humans than in mice, and that this is one of the major causes of infertility. Although the reason is still unknown, investigations into condensin and cohesin may lead to a solution to the riddle.”
Stimulated by the discovery of condensin and cohesin by Hirano, chromosome research has advanced dramatically in recent years. “Considering the fundamental role of the chromosome in life, further research could lead to a series of unexpected discoveries in the years to come.”
Provided by RIKEN