Pull-chain 'polymer' solves puzzle of complex molecular packing
(PhysOrg.com) -- Sometimes the simplest things hold the key to understanding complex effects. It turns out that a humble metal pull-chain -- just like those used on ceiling fans -- can be a pretty good model for complex properties of polymer materials.
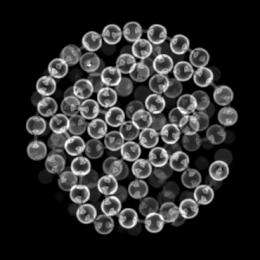
A group of University of Chicago researchers used X-ray microtomography to study what happens when beaded metal chains are packed more and more tightly into a container. The results look similar to those obtained when a polymer becomes rigid, the difference between molten and hard plastic.
Both naturally occurring and synthetic, polymers are large, chain-like molecules found in plastics, rubber and a variety of other substances. With this pull-chain model, the behavior of individual “molecules” can be studied in a way that is impossible with real polymers.
“We were looking for new ways to study how particles pack into stable structures,” said Ling-Nan Zou, PhD’09, lead author of a paper that appeared in the Oct. 16, 2009 issue of Science. “The most dense and stable arrangement is like a pile of oranges at the grocery store, but sometimes less dense arrangements can still be stable, and we don’t really understand why.” These lower-density yet rigid structures are said to be “jammed.”
The researchers chose an elegantly simple physical analog to a polymer: a flexible ball-chain. Each metal sphere is a “monomer,” or polymeric component; the linking rods are “bonds.” When twisted into a loop, there’s a point when the chain stops being flexible: The loop won’t get any smaller. That is, the “bonds” have a maximum angle beyond which they can’t flex. It turns out that this property explains a lot.
To do the experiment, the team cut metal chain into segments of a certain length, poured the segments into a tube, and shook the tube in a defined way to compact the chains. They varied the lengths of the segments and the amount of shaking and then measured the height of the packed chain in the tube, from which they derived the density of the packing.
They also imaged the packed container with x-ray microtomography at beamline 13-BM-D at the U.S. Department of Energy’s Advanced Photon Source at Argonne National Laboratory. The beamline is operated by GeoSoilEnviroCARS, part of the University of Chicago Center for Advanced Radiation Sources.
“We needed the resolution available at this beamline because regular tomography doesn’t work for objects this small,” explained Zou, now a postdoctoral fellow at Harvard University. The spheres in the chains were only 2.4 millimeters in diameter (about the diameter of a grain of rice).
While standard tomography can image sub-millimeter, to accurately trace bonds and bond-angles, the team needed micrometer resolution. “With the data from GSECARS, we can accurately locate every monomer and trace every bond,” Zou said.
They found that even though the density decreased for longer chains, the packings were still stable. The tomography images provided the explanation: The long chains form stiff partial loops (see illustration) that reduce the density but provide stability.
Although the pull-chain or “granular” polymers are clearly different from real polymers in significant respects, there is at least one remarkable similarity. The relation of chain length to density in the granular polymer is nearly identical to the relation of chain length to the liquid-to-glass transition temperature in real polymers.
A mathematical model is being developed based on the new results for granular polymers. “With this kind of model, we may find new behaviors to study experimentally and new ways to explain why polymers have the properties they do,” Zou said. In particular, polymers can behave in a very complex and hard-to-measure fashion under sideways forces, or shear. Now, with the knowledge gained from the experiments on granular polymers, researchers can try to recreate such behavior and then look at the “molecular” level for detailed explanations that could guide the design of new polymer-based materials.
More information: “The Packing of Granular Polymer Chains,” by Ling-Nan Zou, Xiang Cheng, Mark L. Rivers, Heinrich M. Jaeger, and Sidney R. Nagel, Science, Oct. 16, 2009.
Provided by University of Chicago