Nanoscale 'Fountain Pen' Draws Therapeutic Nanodiamonds
A research team at Northwestern University has developed a tool that can precisely deliver tiny doses of drug-carrying nanomaterials to individual cells. The tool, called the nanofountain probe, functions in two different ways. In one mode, the probe acts like a fountain pen with drug-coated nanodiamonds serving as the ink, allowing researchers to create devices by “writing” with it. The second mode functions as a single-cell syringe, permitting direct injection of biomolecules or chemicals into individual cells. The research was led by Horacio Dante Espinosa, Ph.D., and Dean Ho, Ph.D., and the results appear in the journal Small.
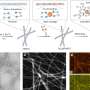
The nanofountain probe could be used both as a research tool for developing next-generation cancer treatments and as a nanomanufacturing tool to build the implantable drug delivery devices that will administer these treatments. In their current work, the investigators use the nanofountain probe to inject tiny doses of nanodiamonds into both healthy and cancerous cells. This technique will help cancer researchers investigate the efficacy of new drug-nanomaterial systems as they become available.
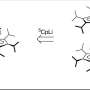
The group also used the same nanofountain probe to pattern dot arrays of drug-coated nanodiamonds directly on glass substrates. The production of these dot arrays, with dots that can be made smaller than 100 nanometers in diameter, provides the proof of concept by which to manufacture devices that will deliver these nanomaterials within the body. The work addresses two major challenges in the development and clinical application of nanomaterial-mediated drug-delivery schemes: dosage control and high spatial resolution.
In fundamental research and development, biologists are typically constrained to studying the effects of a drug on an entire cell population because it is difficult to deliver them to a single cell. To address this issue, the team used the nanofountain probe to target and inject single cells with a dose of nanodiamonds. “This allows us to deliver a precise dose to one cell and observe its response relative to its neighbors,” said Dr. Ho. “This will allow us to investigate the ultimate efficacy of novel treatment strategies via a spectrum of internalization mechanisms.”
Beyond the broad research focused on developing these drug-delivery schemes, manufacturing devices to execute the delivery will require the ability to precisely place doses of drug-coated nanomaterials. Dr. Ho and his colleagues previously developed a polymer patch that could be used to deliver chemotherapy drugs locally to sites where cancerous tumors had been removed. This patch is embedded with a layer of drug-coated nanodiamonds, which moderate the release of the drug. The patch is capable of controlled and sustained low levels of release over a period of months, reducing the need for chemotherapy following the removal of a tumor.
“An attractive enhancement will be to use the nanofountain probe to replace the continuous drug-nanodiamond films currently used in these devices with patterned arrays composed of multiple drugs,” Dr. Ho explained. “This allows high-fidelity spatial tuning of dosing in intelligent devices for comprehensive treatment.” Dr. Espinosa added, “One of the most significant aspects of this work is the nanofountain probe’s ability to deliver nanomaterials coated with a broad range of drugs and other biological agents. The injection technique is currently being explored for delivery of a wide variety of bioagents, including DNA, viruses, and other therapeutically relevant materials.”
This work is detailed in the paper “Nanofountain-probe-based high-resolution patterning and single-cell injection of functionalized nanodiamonds.” An abstract of this paper is available at the journal’s Web site.
Provided by National Cancer Institute (news : web)