Skin biology illuminates how stem cells operate
(PhysOrg.com) -- As a girl, Elaine Fuchs borrowed her mother’s old strainers and mixing bowls to collect polliwogs, an activity she credits for her present-day career as a biologist.
It also helped that her father was a geochemist who studied meteorites, her aunt a radiation expert, her older sister a neuroscientist.
Going on to study science “was almost a no-brainer,” said Fuchs, who did her Ph.D. at Princeton University.
She’s now a professor of cell biology at The Rockefeller University, in New York City, and was at Harvard on March 19 to deliver a lecture on the human organ that has become her life’s work: skin.
Her 50-minute talk, “Skin Stem Cells: Biology and Clinical Promise,” was part of the Dean’s Lecture Series in the sciences sponsored by the Radcliffe Institute for Advanced Study.
Fuchs “recognized very early on that [skin] was a fantastic model system for studying some of the fundamental problems of biology,” said Massachusetts Institute of Technology (MIT) biologist Susan Lindquist, Ph.D. ’76, RI ’08, who introduced Fuchs at the Radcliffe Gymnasium. “For one thing, it’s accessible, and there’s plenty of it.”
Skin is thought to be the largest organ, covering about 18 square feet in the average adult. In every square inch of this protective covering there are a thousand nerve endings and 650 sweat glands.
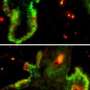
There are also a multitude of hair follicles, the abundant clusters of cells that Fuchs and her investigative team at the Howard Hughes Medical Institute now closely study.
Fuchs is an expert in skin biology — in particular how skin and hair emerge from the same “progenitor” stem cell. That process, she said, is a way to illuminate how stem cells of all types operate.
Her laboratory team investigates how stem cells flower into other kinds of cells, and what happens when the process goes wrong.
The research Fuchs oversees has already provided insight into genetic disorders, cancer, ulcers, and advanced burn therapy. But her lab in Manhattan has one main interest: morphogenesis, the biological process by which organisms grow.
Starting as a postdoc at MIT, Fuchs studied how skin cells build a cytoskeleton, the structural support system that also acts as a conduit for information as the cell grows and divides.
That helped her define ways that skin cells go about changing into other types of cells, and what goes wrong when the cell makes a mistake.
Fuchs built the first mouse model to study human genetic diseases affecting the skin. She studied how cells divide and how they stop dividing — work that gave her insight into cancers of the skin and other organs, when cells multiply uncontrollably.
Her work on skin cell biology and development led Fuchs to advances in treating burns and wounds. It also led to insight into stem cells in general.
She described the skin as a kind of “Saran wrap seal” for the body, an elastic covering that shields muscles, blood vessels, and internal organs. It’s bristling with hair follicles, which push up shafts of dead cells that form into hair — an epidermal appendage that is protective, like feathers in birds or scales on fish.
Skin is also constantly renewing itself, said Fuchs. “Every four weeks you have a brand new surface of your body.”
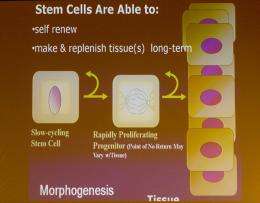
This constant renewal makes the skin one of the body’s chief sources of stem cells, the starter cells that can be transformed into a multiplicity of tissues.
How and why these stem cells get activated in skin is what Fuchs and her team study.
This potential versatility, of course, is what makes stem cells an exciting prospect for future therapies that might replace damaged or missing organ or nerve tissue.
Both “adult” stem cells — those that have a specific function already — and “embryonic” stem cells (ES cells) are self-renewing. They can make and replenish tissues in the long term. But only ES cells, in theory, can generate all 220 cell types present in the human body.
Fuchs, who enlivened her talk with explanatory graphics and snippets of video, showed a clip of a once-paralyzed mouse moving about after its nerve cells had been regenerated through therapy with ES cells.
There are also possible clinical uses for adult skin stem cells, said Fuchs. The cells are already used for burn therapy — a 30-year success story, she said.
Such stem cells might one day be used to treat ulcers by replacing damaged tissue, though the environment of the digestive system is challenging. And there is hope that gene therapy related to stem cells might be used for some skin disorders.
Beyond skin, skin stem cells might be used to prevent blindness by restoring tissue damaged by corneal degenerative diseases.
But Fuchs urged caution about stem cell therapies of any kind. “I don’t want to give people too much optimism with regards to immediate clinical applications,” she said.
More research and clinical work — “quite a few years,” said Fuchs — has to be done in strict and careful scientific cultures like that of the United States.
Prospects for more and better stem cell research have improved with the appearance of a new presidential administration, she said. But until U.S. researchers regain their global footing on the issue of stem cells, they remain “a little voice,” said Fuchs, “trying to stop the world.”
Provided by Harvard University (news : web)