Discovery of ionic elemental crystal against chemical intuition
An ETH Zurich researcher has developed a computational method for predicting the structure of materials. He used it to solve the structure of a newly synthesized form of pure boron that displays some unusual physical properties and brings a surprise: it is partially ionic.
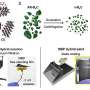
The new structure can be viewed as a NaCl-type structure, with anionic and cationic positions occupied by two different clusters of boron atoms (B12 and B2). The difference of the electronic properties of these clusters brings about charge transfer, making this material a partially ionic boron boride (B2)δ+(B12)δ-. Results have been published in today's "Nature" online magazine.
Boron is the chemical element most sensitive to impurities. This enhanced sensitivity makes experimental studies of this element very difficult. However, with the discovery of a new, superhard phase of the element, the theorists and experimentalists involved in the research have now come a big step closer to understanding boron. A separate publication by the authors in the "Journal of Superhard Materials" demonstrated that the new phase is superhard.
Independently synthesized
The new superhard material was independently synthesized by two researchers who eventually joined forces with crystallographer Artem Oganov's theoretical team. Initially, Jiuhua Chen, a material scientist at Florida International Univer-sity, and Vladimir Solozhenko, a physical chemist at the Centre National de la Recherche Scientifique (CNRS) in France, conducted experiments on extremely pure boron material, containing at most one foreign atom to one million boron atoms. They exposed this material to temperatures of over 1,500 degrees Cel-sius and to pressures in the range 12-30 GPa, similar to those found several hundreds of kilometers inside the Earth. Under these conditions both teams of experimentalists found a new polymorph of boron, but could not solve its struc-ture.
New method leads to breakthrough
Artem Oganov, working at ETH Zurich's Department of Material Science, has now developed a computational method for predicting the stable crystal structures of materials. His calculations reveal that in the new phase, boron atoms form two different kinds of nanoclusters: an icosahedron B12 consisting of twelve atoms and dumbbell B2 consisting of just two boron atoms.
These nanoclusters are arranged in the new phase of boron just as are sodium and chlorine ions in the rock salt (table salt) structure (see diagram). The new phase is predicted to remain stable to 89 GPa. The new knowledge obtained in this study allowed the researchers to propose a phase diagram for boron - the only light element whose phase diagram remained unknown until now.
Unusual properties identified
The unexpected structure of the new phase, which the authors called γ-B, con-tains atoms which are ionized, meaning that the electrons are distributed be-tween the atoms unevenly. According to classical textbooks, ionic bonds are possible only between two different elements, such as sodium and chlorine in table salt. But in the new structure ionic bonds occur between atoms of the same element, though belonging to two kinds of nanoclusters. This ionicity leads to unusual for an element phenomena in dielectric properties, lattice dy-namics, and anomalous electronic properties. Additional experiments carried out by the researchers also show that the new phase is superhard.
Oganov and his colleagues expect that forms of other elements, such as carbon heterofullerites, might display charge transfer and partial ionicity. Now a profes-sor at State University of New York at Stony Brook (USA), Oganov anticipates that sooner or later applications will be developed which are based on ionic elements. These applications could be based on switching on or off the anoma-lous properties (for example, strong infrared absorption) possessed by ionic elements - such properties will display dramatic changes as a result of pres-sure- or temperature-induced phase transitions. In addition, interesting effects related to superconductivity may appear as well.
Paper reference: www.nature.com/nature/journal/ … ull/nature07736.html
Source: ETH Zurich/Swiss Federal Institute of Technology