Electron pairs precede high-temperature superconductivity
(PhysOrg.com) -- Like astronomers tweaking images to gain a more detailed glimpse of distant stars, physicists at the U.S. Department of Energy's (DOE) Brookhaven National Laboratory have found ways to sharpen images of the energy spectra in high-temperature superconductors — materials that carry electrical current effortlessly when cooled below a certain temperature. These new imaging methods confirm that the electron pairs needed to carry current emerge above the transition temperature, before superconductivity sets in, but only in a particular direction.
"Our findings rule out certain explanations for the development of superconductivity in these materials, and lend support to other, competing theories," said Brookhaven physicist Peter Johnson, leader of the group whose work is described in the November 6, 2008, issue of Nature. Homing in on the mechanism for high-temperature (high-Tc) superconductivity may help scientists engineer new materials to make use of the current-carrying phenomenon in transformative applications such as high-efficiency transmission lines in the U.S. power grid.
Scientists already know that electrons in a superconducting material must pair up to carry the current. But whether these pairs form at or above the transition temperature has been a mystery, until now.
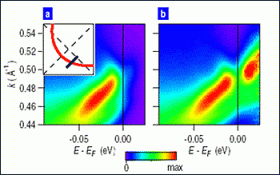
To search for pre-formed electron pairs, the Brookhaven team bombarded a copper-oxide material, held at temperatures above and below the transition temperature, with beams of light from the National Synchrotron Light Source, and analyzed the energy spectrum of electrons emitted from the sample. This method, known as angle-resolved photoemission spectroscopy (ARPES), ordinarily gives a clear picture of only half of the energy spectrum — all the levels electrons can occupy below the so-called Fermi level. To glimpse the other half, above the Fermi level, the scientists employed methods of analysis similar to those used by astronomers to increase the resolution of celestial images.
"If you look through a telescope with poor resolution, you'll see the moon, but the stars are lost," Johnson said. "But if you improve your resolution you see the stars and everything else. By improving our resolution we can use ARPES to see the few electrons that occasionally occupy levels above the Fermi level. We have devised ways to sharpen our images so we can look at the weak signals from above the Fermi level in finer and finer detail."
Seeing both sides of the Fermi level is important because, when a material becomes a superconductor, there is an energy gap surrounding the Fermi level. A perfectly symmetrical gap — equally spaced above and below the Fermi level — is a strong indication that electrons are paired up. That superconducting gap exists at and below the transition temperature, as long as a material acts as a superconductor.
But Johnson's team and other scientists had previously observed a second gap, or pseudogap, in some high-Tc materials, well above the transition temperature. If this pseudogap exhibited the same symmetry around the Fermi level, Johnson reasoned, it would be definitive evidence of paired electrons above the transition temperature. Using their new image-enhancing techniques, Johnson's team demonstrated that the pseudogap does indeed exhibit this same symmetry.
"We can now say for certain that electrons are forming pairs above the transition temperature, before the material becomes a superconductor," Johnson said.
The scientists made another interesting observation: The pairing occurs only along certain directions in the crystalline lattice of atoms making up the material — only along the directions in which copper atoms are bonded with oxygen atoms.
Together, the existence of preformed electron pairs and their directional dependence should help clarify the picture of high-Tc superconductivity, Johnson said. For example, the findings rule out some theories to explain the high-Tc phenomenon (e.g. certain "spin density wave" and "charge density wave" derived theories). But the new findings are consistent with theories that consider the pre-superconducting state to be derived from a "Mott insulator," as well as theories in which "charge stripes," previously discovered at Brookhaven Lab, might play a role in electron pairing.
"It's still a very complicated picture and one of the great mysteries of modern science," Johnson said. "With something like 150 theorists working in the field, we have 150 theories of how these materials work. But as we develop new techniques, we are making progress narrowing down the mechanism."
Provided by Brookhaven National Laboratory