Scientists discover what drives the development of a fatal form of malaria
Platelets – those tiny, unassuming cells that cause blood to clot and scabs to form when you cut yourself – play an important early role in promoting cerebral malaria, an often lethal complication that occurs mostly in children. Affecting as many as half a billion people in tropical and subtropical regions, malaria is one of the oldest recorded diseases and the parasite responsible for it, Plasmodium, among the most studied pathogens of all time. Still, cerebral malaria, which results from a combination of blood vessel and immune system dysfunction, is not well understood.
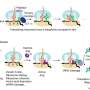
In a study described in the August 14 issue of Cell Host and Microbe, Johns Hopkins researchers reveal that when red blood cells are infected with the malaria parasite, they activate platelets to secrete the PF4 protein, which triggers the immune system to inflame blood vessels and obstruct capillaries in the brain; both are hallmarks of cerebral malaria.
In their experiments, the Hopkins team first infected human red blood cells in culture with the malaria parasite and found that this did, indeed, induce platelet activation.
The researchers then infected separate sets of live mice with the malaria parasite: one set treated so that it lacked platelets altogether and two others treated with aspirin or Plavix, platelet inhibitors that prevent the release of PF4.
The survival rate of mice without platelets as well as those treated with inhibitors was improved over that of the mice left alone, but only when the treatment began very soon after infection. When researchers started treating mice with platelet inhibitors one day after infecting them, those mice survived more often than control mice. However, when researchers waited until after three days to treat infected mice with platelet inhibitors, that group did no better in terms of survival.
"Cerebral malaria is lethal 20 percent of the time in the best of hands, and here we've shown that something as simple as aspirin, because of its affect on platelets, might be able to improve the outcomes of those who contract this deadly form of the disease," says David Sullivan M.D., an associate professor of molecular microbiology and immunology in the Johns Hopkins University Bloomberg School of Public Health.
To make the specific connection between PF4 and malaria, the scientists compared the responses to malaria infection by so-called "wild type" normal mice and mice genetically engineered to lack pF4. They found that the amount of parasite in the blood was the same in both sets of mice. The notable difference was in the animals' immune responses to that same parasite burden. More than 60 percent of the mice lacking PF4 were still alive after day 10, while only 30 percent of the mice with PF4 survived that long.
"The take-home lesson is that platelets, by releasing PF4, are playing an early role in the wind-up phase of cerebral malaria," says Craig Morrell, DVM, Ph.D., an assistant professor of molecular and comparative pathobiology at the Johns Hopkins University School of Medicine. "Our mouse studies show that timing is critical; with the mice, we know when we infected them and controlled when we treated them. A big challenge in translating this to humans is that people don't know when they get infected.
"Platelets don't get any respect, but they're the second most abundant cell in the blood after red blood cells and packed full of factors that rally the immune system to action. By taking what we know about platelets and their activation and applying it to malaria, we have found a driver of cerebral malaria."
Source: Johns Hopkins Medical Institutions