Collaboration helps make JILA strontium atomic clock 'best in class'
A next-generation atomic clock that tops previous records for accuracy in clocks based on neutral atoms has been demonstrated by physicists at JILA, a joint institute of the Commerce Department's National Institute of Standards and Technology and the University of Colorado at Boulder. The new clock, based on thousands of strontium atoms trapped in grids of laser light, surpasses the accuracy of the current U.S. time standard based on a "fountain" of cesium atoms.
JILA's experimental strontium clock, described in the Feb. 14 issue of Science Express, is now the world's most accurate atomic clock based on neutral atoms, more than twice as accurate as the NIST-F1 standard cesium clock located just down the road at the NIST campus in Boulder.
The JILA strontium clock would neither gain nor lose a second in more than 200 million years, compared to NIST F-1's current accuracy of over 80 million years.
The advance was made possible by Boulder's critical mass of state-of-the-art timekeeping equipment and expertise. The JILA strontium clock was evaluated by remotely comparing it to a third NIST atomic clock, an experimental model based on neutral calcium atoms. The best clocks can be precisely evaluated by comparing them to other nearby clocks with similar performance; very long-distance signal transfer, such as by satellite, is too unstable for practical, reliable comparisons of the new generation of clocks. In the latest experiment, signals from the two clocks were compared via a 3.5-kilometer underground fiber-optic cable.
The strontium and calcium clocks rely on the use of optical light, which has higher frequencies than the microwaves used in NIST-F1. Because the frequencies are higher, the clocks divide time into smaller units, offering record precision. Laboratories around the world are developing optical clocks based on a variety of different designs and atoms; it is not yet clear which design will emerge as the best and be chosen as the next international standard. The work reported in Science Express is the first optical atomic clock comparison over kilometer-scale urban distances, an important step for worldwide development of future standards.
"This is our first comparison to another optical atomic clock," says NIST/JILA Fellow Jun Ye, who leads the strontium project. "As of now, Boulder is in a very unique position. We have all the ingredients, including multiple optical clocks and the fiber-optic link, working so well. Without a single one of these components, these measurements would not be possible. It's all coming together at this moment in time."
NIST and JILA are home to optical clocks based on a variety of atoms, including strontium, calcium, mercury, aluminum, and ytterbium, each offering different advantages. Ye now plans to compare strontium to the world's most accurate clock, NIST's experimental design based on a single mercury ion (charged atom). The mercury ion clock was accurate to about 1 second in 400 million years in 2006 and performs even better today, according to Jim Bergquist, the NIST physicist who built the clock. The "best" status in atomic clocks is a moving target.
The development and testing of a new generation of optical atomic clocks is important because highly precise clocks are used to synchronize telecom networks and deep-space communications, as well as for navigation and positioning. The race to build even better clocks is expected to lead to new types of gravity sensors, as well as new tests of fundamental physical laws to increase understanding of the universe. Because Ye's group is able to measure and control interactions among so many atoms with such exquisite precision, the JILA work also is expected to lead to new scientific tools for quantum simulations that will help scientists better understand how matter and light behave under the strange rules governing the nanoworld.
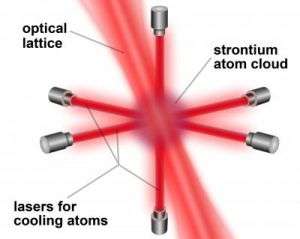
In the JILA clock, a few thousand atoms of the alkaline-earth metal strontium are held in a column of about 100 pancake-shaped traps called an "optical lattice." The lattice is formed by standing waves of intense near-infrared laser light. Forming a sort of artificial crystal of light, the lattice constrains atom motion and reduces systematic errors that occur in clocks that use moving balls of atoms, such as NIST-F1. Using thousands of atoms at once also produces stronger signals and eventually may yield more precise results than clocks relying on a single ion, such as mercury. JILA scientists detect strontium's "ticks" (430 trillion per second) by bathing the atoms in very stable red laser light at the exact frequency that prompts jumps between two electronic energy levels. The JILA team recently improved the clock by achieving much better control of the atoms. For example, they can now cancel out the atoms' internal sensitivity to external magnetic fields, which otherwise degrade clock accuracy. They also characterized more precisely the effects of confining atoms in the lattice.
The NIST calcium clock, which was used to evaluate the performance of the new strontium clock, relies on the ticking of clouds of millions of calcium atoms. This clock offers high stability for short times, relatively compact size and simplicity of operation. NIST scientists believe it could be made portable and perhaps transported to other institutions for evaluations of other optical atomic clocks. JILA scientists were able to take advantage of the calcium clock's good short-term stability by making fast measurements of one property in the strontium clock and then quickly switching to a different property to start the comparison over again.
The JILA-NIST collaborations benefit both institutions by enabling scientists not only to compare and measure clock performance, but also to share tools and expertise. Another key element to the latest comparison was the use of two custom-made frequency combs, the most accurate tool for measuring optical frequencies, which helped to maintain stability during signal transfer between the two institutions.
Citation: A.D. Ludlow, T. Zelevinsky, G.K. Campbell, S. Blatt, M.M. Boyd, M.H.G. de Miranda, M.J. Martin, S.M. Foreman, J. Ye, T.M. Fortier, J.E. Stalnaker, S.A. Diddams, Y. Le Coq, Z.W. Barber, N. Poli, N.D. Lemke, K.M. Beck, & C. Oates. 2008. Sr lattice clock at 1x10-16 fractional uncertainty by remote optical evaluation with a Ca clock. Science Express. Posted online Feb. 14.
Source: National Institute of Standards and Technology





















